On April; 26, 2026, FDA announced that it added 18 compounds to its “Harmful and Potentially Harmful Constituents” (HPHC) list to assess toxicity of tobacco products. This was the first update of the list since it was first released 14 years ago in 2012. The updated list includes several things, most notably propelyne glycol (PG)Continue reading “We support FDA expanding HPHC list and suggest adding more flavoring agents (public comment)”
Author Archives: Stanton Glantz
We support California’s updated”Unflavored Tobacco List” and suggest some improvements
The California Attorney Genera maintains an official “unflavored tobacco product list” to facilitate implementation and enforcement of the state’s flavored tobacco product ban. The AG proposed “readopting” an undated list. We support that proposal and made a few suggestions for improving it. A PDF is available here. This is an excellent model for other states,Continue reading “We support California’s updated”Unflavored Tobacco List” and suggest some improvements”
E-cigarettes increase harm and should be discouraged (new paper)
Pam Ling and I recently published “E-cigarettes increase harm and should be discouraged” in Nature Human Behavior that summarizes the current evidence on e-cigarettes. The Introduction sums up the paper: Ever since they were introduced, arguments that e-cigarettes reduce tobacco harm have hinged on three assumptions: (1) nicotine aerosol from heating a liquid rather thanContinue reading “E-cigarettes increase harm and should be discouraged (new paper)”
FDA should drop its draft guidance to allow some sales of flavored e-cigs because it ignores evidence that harm to youth outweighs theoretical benefits to adults
My colleagues and I just submitted this public comment to FDA opposing its proposal to allow sales of flavored e-cigarettes and other electronic nicotine delivery systems (ENDS) on the grounds that benefits to adults would outweigh harms to kids. It also presents evidence that two-way communication between ENDS and external agencies (that could include ENDSContinue reading “FDA should drop its draft guidance to allow some sales of flavored e-cigs because it ignores evidence that harm to youth outweighs theoretical benefits to adults”
I surprised Michael Smerconsih when he interviewed me about UK generational smoking ban
On April 23, 2026, Michael Smerconsih interviewed me about the UK Generational Smoking ban. I surprised him by arguing that generational smoking bans are bad public policy because they divert resources from more effective tobacco control policies that had faster public health benefits. I am also skeptical that they will be maintained in the longContinue reading “I surprised Michael Smerconsih when he interviewed me about UK generational smoking ban”
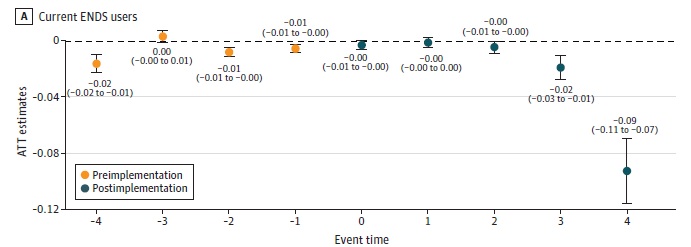
Local flavor bans followed by big drops in youth e-cig use
The evidence that comprehensive flavor bans are followed by drops in tobacco consumption keeps piling up. A new study by Giovanni Appolon, and colleagues, Local Flavored Tobacco Bans and Youth Electronic Nicotine Delivery Systems Use, of 2.8 million California middle and high school students across 483 local jurisdictions found that local flavored tobacco bans thatContinue reading “Local flavor bans followed by big drops in youth e-cig use”
MAHA claims that nicotine is good for you echo decades of tobacco industry propaganda
The New York Times recently highlighted how many “Make America Healthy Again” influencers are promoting the benefits of nicotine, including reversing Alzheimer’s Disease and improving cognition. While we don’t know what role the tobacco companies are playing in this latest resurgence of claims that nicotine is a beneficial substance that poses little or no risk,Continue reading “MAHA claims that nicotine is good for you echo decades of tobacco industry propaganda”
FDA adds 18 chemicals to list of harmful constituents in tobacco products, including propelyene glycol and vegetable glycerine
The FDA is required to maintain a list of “Harmful and Potentially Harmful Constituents” (HPHC) in tobacco products. The original list was released in 2012 and was mostly toxicants in cigarette and other tobacco smoke and smokeless tobacco. In particular, this list did not include important compounds in e-cigarettes and other “new” tobacco products. InContinue reading “FDA adds 18 chemicals to list of harmful constituents in tobacco products, including propelyene glycol and vegetable glycerine”
Evidence that ecigs are a gateway to smoking and promote relapse in former smokers keeps piling up
Jagdish Kaur from WHO and several colleagues from India’s new paper “E-cigarette use and subsequent tobacco smoking initiation: an umbrella review with Bayesian model meta-analysis” provides the most comprehensive summary of evidence that e-cigarettes promote cigarette smoking in nonsmokers available to date. They also show that e-cigarette use in former smokers promotes relapse to smoking.Continue reading “Evidence that ecigs are a gateway to smoking and promote relapse in former smokers keeps piling up”
Nicotine pouches maintain the nicotine market for tobacco companies
Nicotine pouches (like ZYN and on!) are being touted by some advocates as a “harm reduction” alternative to smoking cigarettes. Kelvin Choi and colleagues examined whether this happens in real life in their new paper “Patterns of tobacco product use among US adults who use nicotine pouches and the association between nicotine pouch use andContinue reading “Nicotine pouches maintain the nicotine market for tobacco companies”