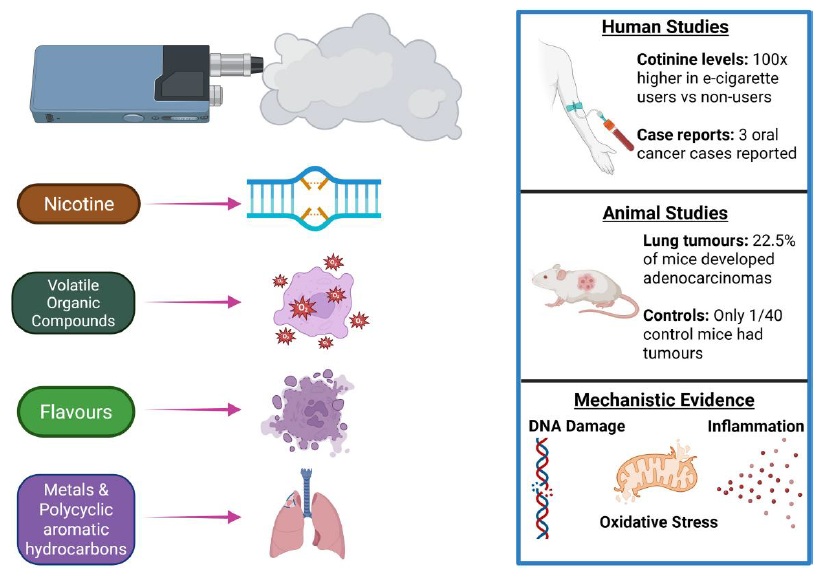
Bernard Stewart and colleagues from Australia just published “The carcinogenicity of e-cigarettes: a qualitative risk assessment” in Carcinogenesis. This review (summary) of the literature cites 116 papers, 15 of which were published in 2025. The fact that so many of the citations are recent is important because the evidence on the health risks of e-cigarettesContinue reading “Broad evidence implicates e-cigarettes as a cause of cancer”
Author Archives: Stanton Glantz
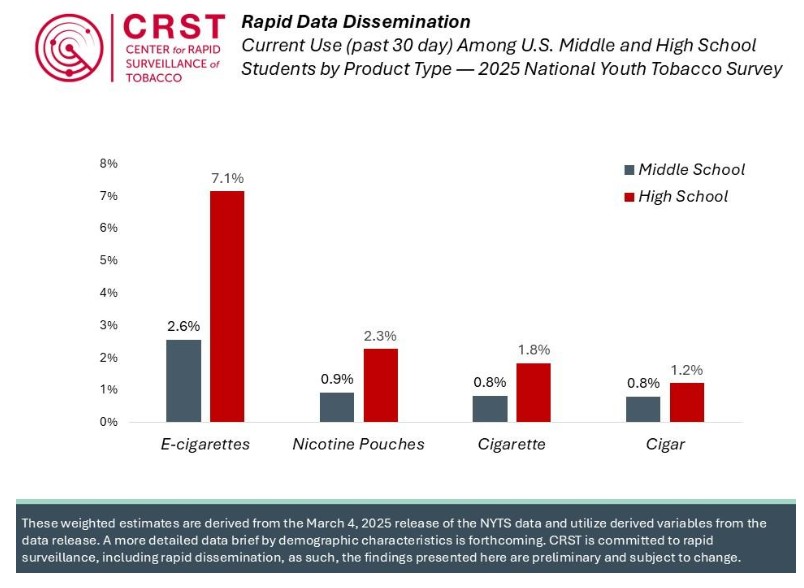
FDA releases 2025 NYTS data without any analysis; Altria and Rutgers (separately) provide summary results
In April 2025, the Trump Administration shuttered the CDC Office on Smoking and Health and stopped collecting data for the 2025 National Youth Tobacco Survey. Later, the Administration announced it was restoring the NYTS and transferring is administration to the FDA Center For Tobacco Products, which had been partnering with CDC since 2021. On MarchContinue reading “FDA releases 2025 NYTS data without any analysis; Altria and Rutgers (separately) provide summary results”
California’s “Unflavored Tobacco List” will improve compliance with and enforcement of flavored tobacco ban
Last year California passed AB 3218 to tighten up and simplify compliance with and enforcement of California’s ban on the sale of flavored tobacco products. Part of this was to have the Attorney General establish and maintain an Unflavored Tobacco List (UTL) that included those tobacco products that could be sold legally in California. toContinue reading “California’s “Unflavored Tobacco List” will improve compliance with and enforcement of flavored tobacco ban”
E-cigarettes are nearly as risky as cigarettes; dual use is worse: The evidence continues to pile up
Using e-cigarettes (vaping) has been promoted as a much safer alternative to smoking cigarettes. However, a new head-to-head comparison of actual disease in e-cigarette users compared to cigarette smokers published in Public Health Reports shows that this assertion is wrong. This new analysis of 124 studies on the association of e-cigarette use with disease outcomesContinue reading “E-cigarettes are nearly as risky as cigarettes; dual use is worse: The evidence continues to pile up”
What the BAT chief scientist had to say about causality half a century ago
E-cigarette advocates, global warming deniers and social media companies continue to sidestep the implications of research showing dangers of their products by, among other things, retreating behind the high walls of claims that the evidence is not “causal.” To put these claims in context, it is worth re-reading the last couple paragraphs from our bookContinue reading “What the BAT chief scientist had to say about causality half a century ago”
DOJ spying on Congress’ viewing of Epstein files echos tobacco company spying on researchers
On February 9, 2026, as required by Epstein Files Transparency Act, the Department of Justice gave members of Congress access to the unredacted Epstein files, but just made four computer terminals at available. This reminded me of a similar situation back in 1999, when the British American Tobacco (BAT) company opened its Guildford Depository toContinue reading “DOJ spying on Congress’ viewing of Epstein files echos tobacco company spying on researchers”
Spinola et al support Brazil’s e-cigarette ban while defending implementation criticism
Shortly before the 11th WHO Framework Convention On Tobacco Control Conference of the Parties in November 2025, Vitoria Borges Spinola, Lucas Porto Santos, Hamilton Roschel, Bruno Gualano, and Megan E. Roberts published “Weak enforcement of Brazil’s E-cigarette ban and the expansion of a dangerous illicit market” in The Lancet Regional Health – Americas arguing thatContinue reading “Spinola et al support Brazil’s e-cigarette ban while defending implementation criticism”
DOJ should have been able to easily release properly redacted Epstein files by December 19, 2025 based on our experience managing the UCSF Industry Documents Library
The issue of redactions – both redacting names of victims and not redacting participants in Jeffery Epstein’s “social circle” — documents has been in the news. There have also been questions about why it took so long for DOJ to release around 3.5 million pages of documents, plus about 2,000 videos and 180,000 images. WorkingContinue reading “DOJ should have been able to easily release properly redacted Epstein files by December 19, 2025 based on our experience managing the UCSF Industry Documents Library”
Congress revives CDC Office on Smoking and Health
Last year the Trump Administration eliminated the CDC Office on Smoking and Health and fired the FDA Center for Tobacco Products leadership, paving the way for FDA to even more closely embrace the tobacco industry’s “harm reduction” marketing messages. OSH ran the effective “Tips from Former Smokers” advertising campaign, supports state health department tobacco controlContinue reading “Congress revives CDC Office on Smoking and Health”
UCSF makes 60,000 more Juul documents public, bringing total to 4.9 million documents so far
The UCSF Industry Documents Library added close to 60,000 Juul Labs documents produced by the Settling States in the multistate litigation against Juul Labs. Files include memos, emails, flavor focus group reports, prospective investor reports, support tickets and more. This brings the total number of Juul Labs files to 4.9 million documents and growing! CheckContinue reading “UCSF makes 60,000 more Juul documents public, bringing total to 4.9 million documents so far”