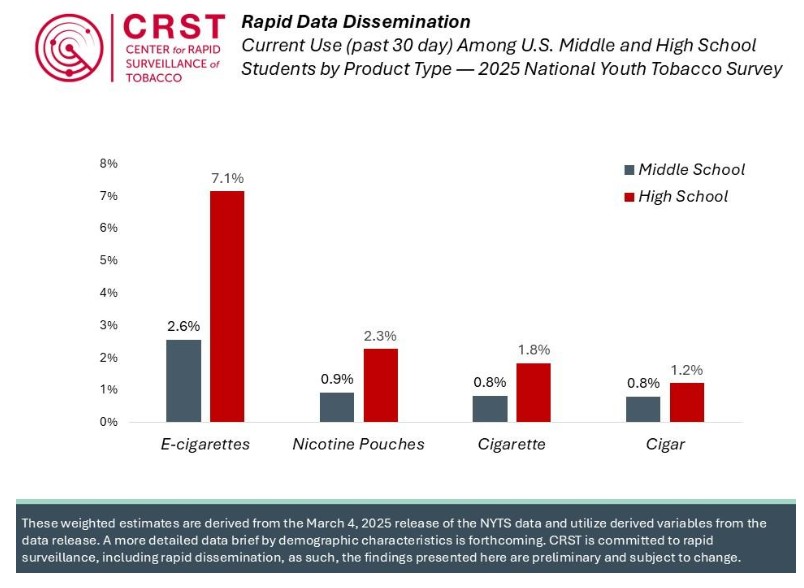
In April 2025, the Trump Administration shuttered the CDC Office on Smoking and Health and stopped collecting data for the 2025 National Youth Tobacco Survey. Later, the Administration announced it was restoring the NYTS and transferring is administration to the FDA Center For Tobacco Products, which had been partnering with CDC since 2021. On MarchContinue reading “FDA releases 2025 NYTS data without any analysis; Altria and Rutgers (separately) provide summary results”
Category Archives: flavors
California’s “Unflavored Tobacco List” will improve compliance with and enforcement of flavored tobacco ban
Last year California passed AB 3218 to tighten up and simplify compliance with and enforcement of California’s ban on the sale of flavored tobacco products. Part of this was to have the Attorney General establish and maintain an Unflavored Tobacco List (UTL) that included those tobacco products that could be sold legally in California. toContinue reading “California’s “Unflavored Tobacco List” will improve compliance with and enforcement of flavored tobacco ban”
UCSF makes 60,000 more Juul documents public, bringing total to 4.9 million documents so far
The UCSF Industry Documents Library added close to 60,000 Juul Labs documents produced by the Settling States in the multistate litigation against Juul Labs. Files include memos, emails, flavor focus group reports, prospective investor reports, support tickets and more. This brings the total number of Juul Labs files to 4.9 million documents and growing! CheckContinue reading “UCSF makes 60,000 more Juul documents public, bringing total to 4.9 million documents so far”
FDA broke the law when it did not allow TPSAC to vote on whether to allow PMI to make modified risk claims for ZYN
The FDA held a meeting of its Tobacco Products Scientific Advisory Committee (TPSAC) on January 22, 2026 to discuss Swedish Match’s application to make the modified risk tobacco product (MRTP) claim for its ZYN flavored nicotine pouches “Using ZYN instead of cigarettes puts you at a lower risk of mouth cancer, heart disease, lung cancer,Continue reading “FDA broke the law when it did not allow TPSAC to vote on whether to allow PMI to make modified risk claims for ZYN”
How e-cigarettes compromise children’s human rights
The new paper “How e-cigarettes compromise children’s human rights” by Tom Gatehouse and colleagues provides more evidence to inform e-cigarette policy making by reminding the delegates to the FCTC Conference of the Parties next week that they need to prioritize protecting kids over any “harm reduction” benefits for adults that the tobacco industry claims forContinue reading “How e-cigarettes compromise children’s human rights”
WHO position on Tobacco Control and Harm Reduction
In the face of a major push by the tobacco companies and their allies to trick Parties to the WHO Framework Convention on Tobacco Control (FCTC) to integrate the industry’s “harm reduction” marketing message into FCTC guidelines, the WHO has issued a position paper on harm reduction (below). The WHO’s bottom line: “When it comesContinue reading “WHO position on Tobacco Control and Harm Reduction”
International Pediatric Association publishes position paper concluding that “e-cigarettes are not an effective harm reduction strategy for adults and pose a serious risk to nicotine naïve youth”
On October 10, 2025, the International Pediatric Association (IPA) published a landmark position paper, “E-Cigarettes and the Nicotine Epidemic: Statement From the International Pediatric Association,” in Pediatrics, the international peer-reviewed journal published by the American Academy of Pediatrics. The paper concluded, “In contrast to early hopes and assumptions, e-cigarettes are not an effective harm reductionContinue reading “International Pediatric Association publishes position paper concluding that “e-cigarettes are not an effective harm reduction strategy for adults and pose a serious risk to nicotine naïve youth””
FDA’s product standard setting a maximum level of nicotine should be expanded to all tobacco products including e-cigs and nicotine pouches
My colleagues at UCSF and Stanford and I have submitted this public comment to the FDA on its prposed nicotine standard urging that the standard be extended to all tobacco products (PDF). The regulations.gov Comment Tracking Number: mfj-v5cz-15yf. The scope of FDA’s proposed tobacco product standard setting a maximum level of nicotine in tobacco productsContinue reading “FDA’s product standard setting a maximum level of nicotine should be expanded to all tobacco products including e-cigs and nicotine pouches”
The evidence for the ecig gateway to smoking keeps piling up; ecig advocates continue to nitpick
Su Golder and colleagues paper “Vaping and harm in young people: umbrella review” sums up the large existing literature on e-cigarettes’ effects on youth, defined as people under 25. In contrast to systematic reviews which find and draw conclusions based on summing up individual studies, this umbrella review is finds and sums up all theContinue reading “The evidence for the ecig gateway to smoking keeps piling up; ecig advocates continue to nitpick”
Our public comment supporting creation of Calif Unflavored Tobacco List with some suggestions for improvements
I previously suggested people provide public comments supporting the California Attorney General’s procedures to create an Unflavored Tobacco Products List to facilitate implementation and enforcement of California’s flavored tobacco product ban and indicated our preliminary ideas. You can read all 24 comments that were submitted here. All support continuing the survey; about half the commentsContinue reading “Our public comment supporting creation of Calif Unflavored Tobacco List with some suggestions for improvements”