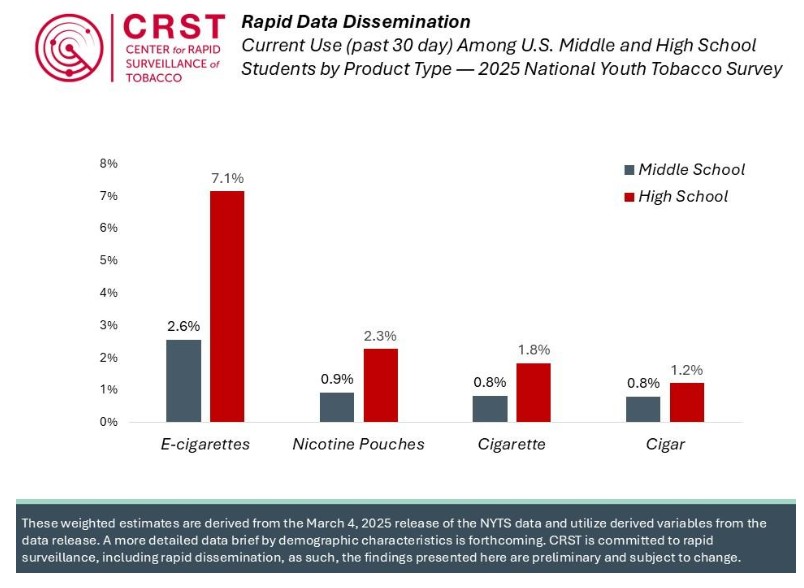
In April 2025, the Trump Administration shuttered the CDC Office on Smoking and Health and stopped collecting data for the 2025 National Youth Tobacco Survey. Later, the Administration announced it was restoring the NYTS and transferring is administration to the FDA Center For Tobacco Products, which had been partnering with CDC since 2021. On MarchContinue reading “FDA releases 2025 NYTS data without any analysis; Altria and Rutgers (separately) provide summary results”
Category Archives: FDA
E-cigarettes are nearly as risky as cigarettes; dual use is worse: The evidence continues to pile up
Using e-cigarettes (vaping) has been promoted as a much safer alternative to smoking cigarettes. However, a new head-to-head comparison of actual disease in e-cigarette users compared to cigarette smokers published in Public Health Reports shows that this assertion is wrong. This new analysis of 124 studies on the association of e-cigarette use with disease outcomesContinue reading “E-cigarettes are nearly as risky as cigarettes; dual use is worse: The evidence continues to pile up”
FDA broke the law when it did not allow TPSAC to vote on whether to allow PMI to make modified risk claims for ZYN
The FDA held a meeting of its Tobacco Products Scientific Advisory Committee (TPSAC) on January 22, 2026 to discuss Swedish Match’s application to make the modified risk tobacco product (MRTP) claim for its ZYN flavored nicotine pouches “Using ZYN instead of cigarettes puts you at a lower risk of mouth cancer, heart disease, lung cancer,Continue reading “FDA broke the law when it did not allow TPSAC to vote on whether to allow PMI to make modified risk claims for ZYN”
RFK Jr strongly endorses tobacco industry harm reduction argument for ecigs and nicotine pouches
In a video interview circulated by Danny Gillis owner of SevierVapor and Executive Director Tennessee Smoke Free Association, Health and Human Services Secretary Robert F. Kennedy, Jr strongly endorses e-cigarettes and nicotine pouches for “harm reduction.” He also cites industry arguments that nicotine helps people with Alzheimer’s disease and dimentia (at 50 seconds in theContinue reading “RFK Jr strongly endorses tobacco industry harm reduction argument for ecigs and nicotine pouches”
Plain language summary of meta-analysis of ecigs and disease available
Last year we published “Population-Based Disease Odds for E-Cigarettes and Dual Use versus Cigarettes” that showed that some disease risks of e-cigarettes are indistinguishable from cigarettes and for others they on only slightly lower. This paper continues to attract interest, so I prepared a one page plain language summary, together with some frequently asked questions.Continue reading “Plain language summary of meta-analysis of ecigs and disease available”
FDA Tobacco Products Scientific Advisory Committee should recommend against FDA allowing Philip Morris Products continuing to make misleading reduced risk claims about IQOS
FDA has scheduled a virtual meeting of its Tobacco Products Scientific Advisory Committee (TPSAC) meeting for October 7, 2025, to discuss the modified risk tobacco product (MRTP) renewal applications submitted by Philip Morris Products S.A. for several IQOS heated tobacco products that is happening despite the government shutdown. (Participants can attend via a live webcast.)Continue reading “FDA Tobacco Products Scientific Advisory Committee should recommend against FDA allowing Philip Morris Products continuing to make misleading reduced risk claims about IQOS”
FDA’s product standard setting a maximum level of nicotine should be expanded to all tobacco products including e-cigs and nicotine pouches
My colleagues at UCSF and Stanford and I have submitted this public comment to the FDA on its prposed nicotine standard urging that the standard be extended to all tobacco products (PDF). The regulations.gov Comment Tracking Number: mfj-v5cz-15yf. The scope of FDA’s proposed tobacco product standard setting a maximum level of nicotine in tobacco productsContinue reading “FDA’s product standard setting a maximum level of nicotine should be expanded to all tobacco products including e-cigs and nicotine pouches”
FDA setting a maximum level of nicotine in tobacco products without exceptions for HTP and waterpipe will improve health
My colleagues at UCSF and Stanford and I have submitted this public comment to the FDA supporting a product standard limiting nicotine in tobacco products and opposing exceptions for heated tobacco products and waterpipe (PDF). The Comment Tracking Number in regulations.gov is mfj-vc5x-sbgb .FDA’s proposed tobacco product standard setting a maximum level of nicotine inContinue reading “FDA setting a maximum level of nicotine in tobacco products without exceptions for HTP and waterpipe will improve health”
UCSF launches new improved Industry Documents Library
Today the UCSF Industry Documents Library launched a complete redesign of the website that aligns with evolving institutional and user research needs and delivers a new and improved user experience. What’s new? Save the date – On Tuesday, September 9, 1:30 p.m. ET/10:30 a.m. PT UCSF is hosting a live website open house and Q&AContinue reading “UCSF launches new improved Industry Documents Library”
Support CDC plan to reinstate NYTS by Aug 15 deadline to submit supportive public comments
In April 2025, the Trump Administration pulled the plug on the National Youth Tobacco Survey in the middle of collecting the 2025 sample, essentially blinding the federal government and the public from important detailed information on youth tobacco use. Now, they are proposing to reinstate it. If implemented properly, this is an important decision. ItContinue reading “Support CDC plan to reinstate NYTS by Aug 15 deadline to submit supportive public comments”