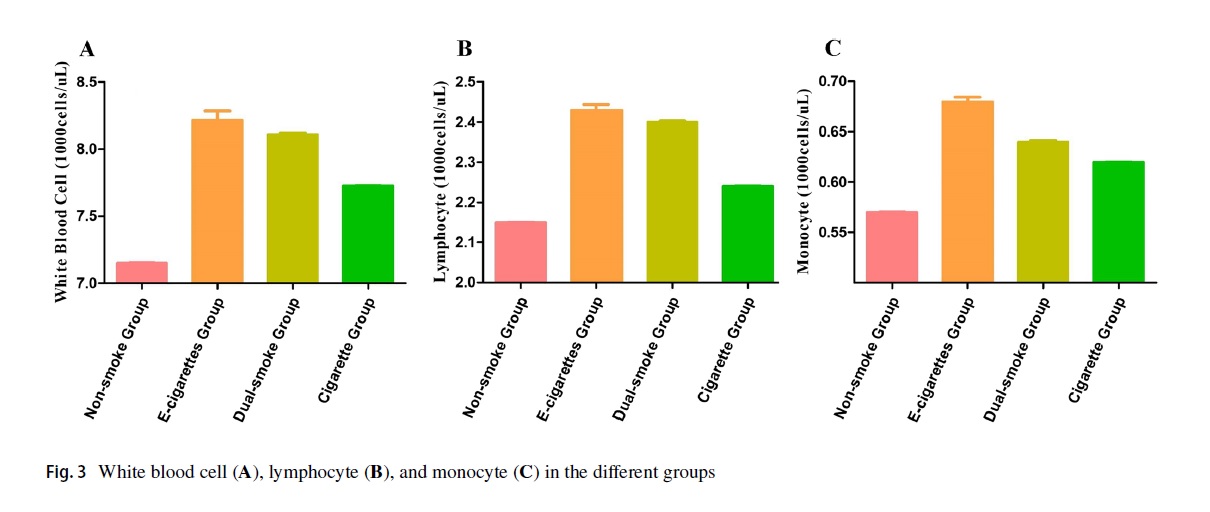
Yao Wang and colleagues compared the results of the Complete Blood Count (CBC), a routine blood tests used in clinical practice to diagnose a range of conditions, among participants in the US National Health and Nutrition Examination Survey (NHANES). In contrast to most national health survey, NHANES includes in person clinical tests on participants. TheirContinue reading “Another organ e-cigs adversely affect: blood”
Category Archives: vaping
FDA science leader Matt Holman leaves FDA for PMI
Matt Holman, who has served as head of the FDA Center for Tobacco Products since 2017, is leaving the FDA to take a job with Philip Morris International. According to a story in The Hill, a spokesperson for Philip Morris said Holman “is committed to helping existing adult smokers access scientifically substantiated smoke-free alternatives whileContinue reading “FDA science leader Matt Holman leaves FDA for PMI”
Since FDA has made a mess of Juul, the health community & Congress should force FDA to ban menthol e-cigs now
The day after FDA denied Juul’s application to continue selling its e-cigarettes on June 23, 2022, Juul sued and obtained an emergency Stay from the US Court of Appeals. On July 5, the FDA announced that it was issuing its own Administrative Stay and allowed Juul to remain on the market while the FDA considersContinue reading “Since FDA has made a mess of Juul, the health community & Congress should force FDA to ban menthol e-cigs now”
White House OKs FDA moving forward on reducing nicotine in cigs; FDA can and should ban menthol ecigs now
On June 21, 2022, the Biden Administration announced that FDA would be moving forward to develop a product standard limiting nicotine in cigarettes and other combusted tobacco products to nonaddictive levels. FDA has been formally considering such a rule for a long time. In 2018, FDA solicited public comment on such a rule. At thatContinue reading “White House OKs FDA moving forward on reducing nicotine in cigs; FDA can and should ban menthol ecigs now”
Direct evidence that Juul increases nicotine addiction, especially among kids
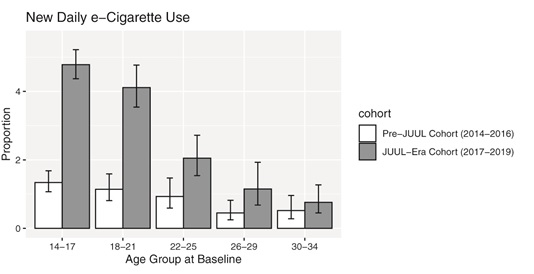
John Pierce and his colleagues at UCSD just published “Daily E-cigarette Use and the Surge in JUUL Sales: 2017-2019” that shows shows that after Juul came on the market there was a 3.6-fold higher rate of progression to daily e-cigarette use — a marker of addiction — among 14-17 year olds compared to 3 yearsContinue reading “Direct evidence that Juul increases nicotine addiction, especially among kids”
FDA prejudged e-cigs as good before it evaluated a single application
An August 31, 2020 memo detailing FDA’s plan for dealing with the expected flood of Premarket Tobacco Applications (PMTAs), clearly prejudges e-cigarettes as a good thing before looking at a single application. In particular, the FDA describes its “public heath goals” as including Ensure a variety of ENDS [electronic nicotine delivery systems] have an opportunityContinue reading “FDA prejudged e-cigs as good before it evaluated a single application”
FDA does JT/Logic’s work for it to justify authorizing 3 of its ENDS
On March 24, 2022 FDA authorized the sale of three electronic nicotine delivery systems distributed by Japan Tobacco’s US company Logic Technology Development, – two conventional e-cigarettes and one heated tobacco product. FDA withheld the Technical Project Lead (TPL) report that provides the scientific justifications for its decision. Thanks to my colleague Lauren Lempert filingContinue reading “FDA does JT/Logic’s work for it to justify authorizing 3 of its ENDS”
FDA proposed updating its HPHC list in 2019; it needs to finish the job
One of the nerdy, but very important, parts of the FDA tobacco product regulatory process is the list of “Hazardous and Potentially Hazardous Compounds” (HPHC) list that FDA released in 2012. This list contains 93 bad chemicals — mostly carcinogens — that FDA identified as important when assessing the risks of tobacco products. It hasContinue reading “FDA proposed updating its HPHC list in 2019; it needs to finish the job”
FDA’s proposed ban on menthol cigarettes explains why FDA needs to reverse its de facto approvals of menthol e-cigarettes
The FDA’s decisions to act on premarket tobacco product applications (PMTAs) for e-cigarettes has been explicit about two points: (1) it is authorizing tobacco flavors because kids don’t seem to use tobacco flavored e-cigs and (2) it is blocking flavored e-cigs – except menthol – because kids like flavors. Because they can’t justify allowing mentholContinue reading “FDA’s proposed ban on menthol cigarettes explains why FDA needs to reverse its de facto approvals of menthol e-cigarettes”
FDA authorizes two more RJR tobacco e-cigs and lets menthol remain on the market, ignoring its own conclusion that menthol reinforces nicotine addiction in kids’ developing brains
Today (May 12, 2022) FDA announced that it was authorizing the sale of two additional RJR e-cigarettes, Vuse Vibe (a vape pen) and Vuse Ciro (similar to previously authorized Vuse Solo). FDA didn’t act on RJR’s most popular Vuse product, Vuse Alto, so it continues on the market. Following FDA’s de facto policy, both wereContinue reading “FDA authorizes two more RJR tobacco e-cigs and lets menthol remain on the market, ignoring its own conclusion that menthol reinforces nicotine addiction in kids’ developing brains”