The FDA’s decisions to act on premarket tobacco product applications (PMTAs) for e-cigarettes has been explicit about two points: (1) it is authorizing tobacco flavors because kids don’t seem to use tobacco flavored e-cigs and (2) it is blocking flavored e-cigs – except menthol – because kids like flavors. Because they can’t justify allowing menthol e-cigs on scientific or public health grounds, they have allowed them to say on the market by simply not acting on the applications for menthol e-cigs.
Why? FDA leadership has adopted the tobacco industry’s “continuum of risk” framing and repeatedly said that the fundamental goal of the FDA is moving people down the continuum of risk, which, the FDA assumes, requires menthol e-cigarettes (and other “potentially reduced risk” products) to be available to encourage cigarette smokers to “switch.”
FDA’s proposed rule banning menthol cigarettes forced the its hand because FDA has already authorized the sale of Philip Morris’ IQOS Marlboro Smooth Menthol and Marlboro Fresh Menthol HeatSticks and 22nd Century Group’s Moonlight Menthol Very Low Nicotine cigarettes.Because these products are technically cigarettes, the already-authorized menthol versions prohibited under the new rule.
To protect these products, the FDA was forced to propose an exception to the menthol rule so it could keep these products on the market. While applying to these two products, the logic of the exemption explains how FDA justifies (at least to itself) leaving menthol e-cigarettes on the market.
| NOTE: This is a long blog post, but most of the length is simply quoting the FDA’s proposed menthol rule back to the FDA. All the quotes are from the FDA’s proposed rule ending menthol cigarettes. |
A careful reading of the FDA’s menthol cigarette rule, however, does just the opposite: It makes a compelling case for FDA to deny the pending menthol e-cigarette PMTA applications now.
The FDA summarizes its position on “potentially reduced risk products” when it proposes creating exemptions for the “cigarette” products listed above, this is almost certainly why it is allowing menthol e-cigarettes the remain on the market without explicitly justifying its action.
3. Given the Harmful Effects of Cigarette Smoking, Switching to a Potentially Less Harmful Nicotine Delivery Product May Reduce Death and Disease
FDA recognizes that smokers who choose to switch completely to a potentially less harmful nicotine delivery product to maintain their nicotine dose also could, to the extent that those products result in less harm, significantly reduce their risk of tobacco-related death and disease (Ref. 271). The least harmful nicotine delivery products available to smokers are the pharmaceutical nicotine replacement therapies already approved by FDA as both safe and effective cessation tools, many of which are available in a variety of flavors, including mint, which could appeal to menthol smokers. However, smokers may also transition to tobacco products which utilize other forms of nicotine delivery in place of smoking combusted cigarettes. These include smokeless tobacco, dissolvable products, and ENDS products, among others. (page 83)
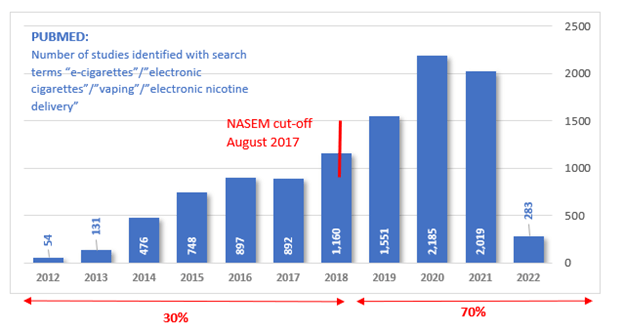
The scientific evidence the FDA cites here (and at other places in the proposed rule to support its position) is the outdated 2018 National Academies of Sciences, Engineering, and Medicine report, “Public Health Consequences of E-Cigarettes” [1] (Ref 271). While this report was a reasonable summation of the evidence five years ago (as of August 31, 2017), of the more than 10,000 articles indexed on PubMed mentioning e-cigarettes or vaping, more than two-thirds were published after then [2]. FDA needs to update its thinking to reflect all this new information.
The FDA should stop speculating about potential benefits of e-cigarettes and base its decision making on actual demonstrated effects
Talking about “potential” benefits was reasonable when e-cigarettes first came on the market and not enough time had passed for the benefits to be manifest, but we are well past that point. E-cigarettes have been sold in the US for 16 years, since 2006. As the FDA carefully describes in the menthol rule, many of the benefits of smoking cessation (drops in heart and lung disease) accrue quickly, and even the “slow” ones (like reduction in cancer risk) take much less than 16 years to manifest themselves.
Here is what FDA says in the proposed menthol rule:
The benefits associated with smoking cessation happen quickly (Ref. 250). Within 2 to 12 weeks of quitting smoking, an individual’s lung function and blood circulation improve (Ref. 250). During the first 1 to 9 months after cessation, coughing and shortness of breath decrease (Ref. 250). Within several months of quitting smoking, individuals can expect improvement in lung function (Ref. 250).
The benefits continue for those who remain smoke-free. Smoking cessation reduces the risk of cancers and other diseases (Ref. 245). For example, the risk of fatal lung cancer in adults over 55 is about 25 times higher among smokers relative to people who have never smoked (Ref. 251). After 10-15 years of abstinence from smoking, the risk of lung cancer is about 50 percent of the risk for individuals who continue to smoke (Ref. 245). The risk of cancer of the mouth, throat, esophagus, stomach, bladder, cervix, pancreas, liver, kidney, colon, rectum, and the risk of acute myeloid leukemia also decreases (Refs. 252 and 245). The evidence is also sufficient to infer that the risk of stroke decreases after smoking cessation, and approaches that of never smokers over time (Ref. 245). Furthermore, the evidence is sufficient to infer that the relative risk of coronary heart disease among former smokers compared with never smokers falls rapidly after cessation and then declines more slowly (Ref. 245). (page 79-80)
It’s true that the FDA does not claim e-cigarettes are risk free: but FDA assumes – without providing any actual evidence to support its assumption – that e-cigarettes are 85% safer than cigarettes. (They do cite a single paper, Reference 46 [3], but that paper does not cite any actual evidence to support the 85% safer number.)
And FDA ignores thousands of papers showing substantial risks.
The simple reality is that if e-cigarettes actually reduced population-level risks it would be obvious by now. It’s not.
FDA needs to accurately engage the full scientific literature
FDA sums up its case for exempting some menthol tobacco products from its proposed ban
In surveys, some menthol cigarette smokers and some dual users of menthol cigarettes and ENDS [electronic nicotine delivery systems, which includes e-cigarettes and heated tobacco products] report intending to use ENDS if menthol cigarettes were no longer available (Refs. 221, 272, and 222). Experimental marketplace studies also suggest that, in addition to taking other actions, some menthol smokers may switch partially or fully to ENDS in the event of a menthol cigarette ban (Refs. 273 and 225). These empirical findings are consistent with the 2020 Surgeon General’s Report, titled “Smoking Cessation,” and several systematic reviews, which suggest that some adult cigarette smokers report using ENDS to try to reduce or quit smoking (Refs. 245, 274-276). The literature also suggests that cigarette smokers who use ENDS more frequently (versus less frequently) have improved success in switching, however the long-term patterns of use remain unknown (Refs. 271, 277-279). (page 83)
This statement has a lot of problems.
Reference 274 is our meta-analysis of e-cigarettes and adult smoking cessation [4]. The point of our meta-analysis is not that “some adult cigarette smokers report using ENDS to try to quit smoking,” but what the effect of that decision is. The statement “The literature also suggests that cigarette smokers who use ENDS more frequently (versus less frequently) have improved success in switching” is correct but misleading. We also found that smokers who use e-cigarettes less than daily (most users) are significantly less likely to quit. The overall effect among all smokers is null (i.e., no effect on having stop smoking).
The FDA ignores this point.
The FDA also ignores another large recent meta-analysis that finds no quitting benefit of E cigarettes [5].
Another problem with the FDA’s argument is that three of the cited studies (Ref 271, 275 and 276) are randomized clinical trials (RCTs), which are relevant to e-cigarettes as medicines, not consumer products. In any event, because the FDA Center for Tobacco Products cannot regulate how e-cigarettes as consumer products are used, the fact that daily users quit more is not relevant.
Ref 245 is the 2020 Surgeon General Report on smoking cessation [6]. The Surgeon General does not conclude that e-cigarettes are effective for smoking cessation. Its main conclusion is: “Therefore, it is important to continue (a) monitoring the findings of research on the potential of e-cigarettes as a smoking cessation aid and (b) evaluating the positive and negative impacts that these products could have at the individual and population levels, so as to ensure that any potential benefits among adult smokers are not offset at the population level by the already marked increases in the use of these products by youth.”
The meta-analyses answer this question. The answer is: No.
More important, the long-term patterns of use are known. The FDA fails to discuss two strong long-term (3 year) longitudinal studies of over 3500 adult smokers who purposefully used e-cigarettes to quit. Both show that smokers who use e-cigarettes to quit are substantially less likey to quit cigarettes that smokers who don’t use e-cigarettes.
The first [7], Longitudinal transition outcomes among adult dual users of e-cigarettes and cigarettes with the intention to quit in the United States: PATH Study (2013-2018, found that smokers who used e-cigarettes to quit were 83% and 79% less likely to transition to cessation or mono cigarette use that smokers who did not use e-cigarettes. Similarly, the second [8], Effectiveness of e-cigarettes as aids for smoking cessation: evidence from the PATH Study cohort, 2017-2019, found that cigarette abstinence for e-cigarette users (9.9%) was lower than for no e-cigarette product use (18.6%). (Conventional FDA approved cessation therapies also worked better than e-cigarettes.) They concluded, “Sales increases in high nicotine e-cigarettes in 2017 did not translate to more smokers using these e-cigarettes to quit smoking. On average, using e-cigarettes for cessation in 2017 did not improve successful quitting or prevent relapse.”
In addition, a meta-analysis of the effects of e-cigarettes on relapse to smoking among former smokers [9] concluded former smokers who used e-cigarettes were significantly more likely to relapse to smoking than former smokers who did not use e-cigarettes. Long-term former smokers were the main contributors for the higher relapse risk. Ignoring this effect is another serious problem in the FDA’s analysis. This finding is particularly relevant in the context of menthol, because the FDA makes a good case that eliminating menthol would reduce relapse.
In short, a comprehensive assessment of the current state of the science is that at best e-cigarettes do not help smokers quit and, more likely, make it harder to quit and promote relapse among former smokers.
Absent quitting benefits, the FDA’s “off ramp” theory evaporates.
The evidence FDA presents from actual implementations of bans on sales of menthol cigarettes does not support FDA’s assumption that menthol e-cigarettes are required for current menthol cigarette smokers
Most directly, FDA cites research from Ontario, Canada after it banned the sale of menthol cigarettes, but exempted menthol e-cigarettes. FDA wrote:
Data from the 2017 Ontario menthol sales restriction did not show increases in menthol smokers’ self-reported use of e-cigarettes (Ref. 280) or increases in retail sales of e-cigarettes (Ref. 231) following policy implementation. To the extent that this may occur following implementation of this product standard, FDA recognizes that completely switching from combusted tobacco products to ENDS has the potential to reduce some tobacco-related disease risks among individual users (Ref 271). (page 84)
There are three important points to be made about this paragraph:
- The first sentence notes that in Ontario where there was also a restriction on sales of menthol cigarettes but not menthol e-cigarettes, menthol smokers did not switch to menthol e-cigarettes.
- The second sentence does not make any sense because menthol smokers did not switch to menthol e-cigs.
- This finding is actually evidence against the need for menthol e-cigarettes and an exemption to allow menthol in other cigarette products.
FDA’s assessment of what happened in Minneapolis when it enacted a comprehensive flavor ban provides more evidence against the need for a “menthol e-cigarette escape hatch.”
In recent years, several U.S. localities and some states have placed restrictions on the sale of menthol cigarettes in addition to restrictions on the sale of other flavored tobacco products. Results from evaluations of these policies provide evidence of decreases in use and sales of tobacco products after policy implementation (Refs. 190-193). In 2018, Minneapolis and St. Paul, Minnesota, expanded their sales restrictions on flavored tobacco products (including e-cigarettes) to include menthol, mint, and wintergreen tobacco products. An evaluation of this sales restriction found decreases in youth cigarette (3.8 percent to 2.3 percent), cigar (2.7 percent to 1.6 percent), smokeless tobacco (1.6 percent to 1.2 percent), and hookah (2.4 percent to 1.3 percent) product use after policy implementation in the Twin Cities metro area, which includes Minneapolis and St. Paul (Ref. 192). An increase in youth e-cigarette prevalence from 10.5 percent to 15.7 percent occurred after the policy in the Twin Cities, but this increase was lower than the rest of the State of Minnesota where e-cigarette prevalence increased from 10.0 percent to 18.8 percent (Ref. 192). Although prevalence of youth overall tobacco use increased after the policy in the Twin Cities from 12.2 percent to 16.5 percent and increased in the rest of the State from 13.9 percent to 20.1 percent, these increases were driven by youth e-cigarette use and align with national youth tobacco use trends (Ref. 192). Importantly, the increases in youth overall tobacco use after the policy were lower in the Twin Cities than in the rest of the State, suggesting that the policy mitigated increases in overall tobacco use. (page 56-57)
This is more evidence against the need to keep menthol e-cigarettes and supporting a menthol rule with no exceptions.
FDA also cites San Francisco as evidence that comprehensive flavor sales bans work:
In July 2018, San Francisco, California, implemented a sales restriction on all flavored tobacco products, including menthol cigarettes. The San Francisco Department of Public Health announced that enforcement would begin January 2019 and enforcement with routine retailer compliance inspections began April 2019 (Ref. 194). An evaluation of the impact of the San Francisco policy on tobacco product sales, a proxy for consumption, found that total tobacco sales decreased by a statistically significant 25 percent from before policy implementation (July 2015-July 2018) to a post-policy enforcement period (January-December 2019) (Ref. 190). This study also found a statistically significant decrease in the overall sales of flavored tobacco products (from 39,350 average weekly unit sales to 1,546 average weekly unit sales), including menthol cigarettes (from 21,463 average weekly unit sales to 860 average weekly unit sales), to low levels after policy enforcement (Ref. 190). Findings that total tobacco sales and flavored tobacco sales decreased post policy suggest that consumers did not completely substitute non flavored tobacco products for flavored tobacco products, and that such a policy can be implemented effectively and reduce sales of products as intended.
Changes in sales of tobacco products in San Francisco after policy enforcement were also reflected in young adult tobacco use patterns. A retrospective study of a convenience sample of young adult ever tobacco users in San Francisco found a statistically significant lower prevalence of overall tobacco use among 18-to 24-year-olds (from 100 percent to 82.3 percent) and 25-to 34-year-olds (from 100 percent to 92.4 percent) about 11 months after policy enforcement (November 2019) (Ref. 191). (page 58)
FDA’s discussion of Massachusetts’ 2020 comprehensive sales ban on flavored tobacco products (which includes menthol e-cigarettes) was followed by big drops in cigarette smoking (page 53).
FDA summarizes the experience in Lowell Massachusetts and found that “overall, youth did not switch to non-flavored or menthol tobacco products, and that the policy help to reduce the use of tobacco products among youth (reference 201)” despite the fact that menthol was excluded from the policy (page 60 – 61).
In contrast to all this empirical evidence based on actual experience showing no need to exempt menthol e-cigarettes, FDA relies heavily on an “expert elicitation study” by Levy et al (ref 211 [10]):
FDA expects that, if this proposed rule is finalized and menthol is prohibited as a characterizing flavor in cigarettes, many menthol cigarette smokers will either quit smoking or switch to a non-combusted tobacco product, such as ENDS. In an expert elicitation study estimating transitions in use under both menthol ban and status quo scenarios, the panel of experts estimated that an additional 20.1 percent of menthol smokers ages 35 to 54 would cease combustible tobacco use over 2 years under a menthol ban compared to the status quo, with about half (10.3 percent) switching to ENDS and about half (10 percent) quitting all tobacco use (Ref. 211). The expert panel also estimated that an additional 30.1 percent of menthol smokers ages 18 to 24 would cease combustible tobacco use over 2 years, with 15.6 percent switching to ENDS and 12.3 percent quitting all tobacco use (Ref. 211). Some menthol cigarette smokers may switch to non-menthol cigarettes. The expert elicitation study suggested that among menthol smokers age 35 to 54, 45.7 percent would become non-menthol cigarette smokers (compared to 4.6 percent under the status quo) while 3.7 percent would become non-menthol cigar smokers (compared to no change under the status quo) (Ref. 211). The expert elicitation study and the resulting population modeling study, which utilized the expert elicitation, are discussed in further detail in section V.C.5 of this document. (page 65-44)
This paper was submitted in late 2020, before most of the actual empirical evidence on what happens when sales of flavored tobacco products are banned was published. In addition, the transitions that menthol smokers were estimated to make were: “Experts considered a range of transitions that included instead initiating into (1) nonmenthol cigarettes, (2) nonmenthol cigars (including little cigars), (3) illicit menthol cigarettes or cigars,29 (4) smokeless tobacco, (5) novel nicotine delivery products (NNDPs, such as e-cigarettes and heated tobacco products), and (6) no regular tobacco product (cigarette, cigar, smokeless, or NNDP) use.” Note that the NNDP group did not specify menthol NNDPs. The FDA implicitly makes that assumption later when using ref 211 to justify the need for their exemption.
In any event, Ref 211 is now obsolete, since FDA has presented ample direct empirical evidence that is more reliable than “expert opinion.” And that empirical evidence does not document any need for menthol e-cigarettes or exceptions in the menthol cigarette ban.
All the evidence FDA presents demonstrating that menthol interacts with nicotine to increase addiction in youth applies to all tobacco products, not just cigarettes
FDA’s strong justification for prohibiting menthol in cigarettes grows out of the biological effects of menthol and how it amplifies the addictive effects of nicotine, particularly in youth. The fact that the aerosol used to deliver the menthol/nicotine mixture is generated by combustion in a cigarette does not matter. There is no evidence that combustion changes menthol or nicotine in a way that affects any of these processes. This evidence also requires banning menthol e-cigarettes.
The beginning of section IIIA of the menthol cigarette ban provides a good succinct summary, together with references, of the scientific evidence on the adverse effects of menthol on health:
Menthol is a flavor compound that is added to cigarettes, which produces a minty taste and cooling sensation when inhaled (Ref. 2). These sensory properties contribute to smoker perceptions that menthol cigarettes are easier to inhale, are less irritating, have a better taste, are smoother and more refreshing than non-menthol cigarettes (Refs. 3-5). Menthol’s flavor and sensory effects reduce the harshness of cigarette smoking among new users and facilitate experimentation and progression to regular smoking of menthol cigarettes, particularly among youth and young adults (Refs. 6-7, 5, 8). As a result, the brain is repeatedly exposed to nicotine and susceptible to nicotine addiction (Ref. 9). …
In addition to its flavor and sensory effects, menthol contributes to a greater risk of
nicotine dependence by enhancing the addictive effects of nicotine in the brain by affecting mechanisms involved in nicotine addiction (Refs. 10-13). Clinical data show that menthol cigarette smokers have higher levels of brain nicotinic receptors compared to non-menthol smokers (Ref. 14). Studies demonstrate that menthol, like nicotine, binds to nicotinic receptors in the brain (Refs. 15 and 16), and menthol alone can increase the number of nicotinic receptors in the brain (Refs. 10 and 11). Evidence demonstrates that the combined effects of menthol and nicotine in the brain are associated with behaviors indicative of greater addiction to nicotine compared to nicotine alone (Refs. 10 and 12).
Youth and young adults are particularly susceptible to becoming addicted to nicotine. Due to its ongoing development, the adolescent brain, which continues to develop until about age 25, is more vulnerable to nicotine’s effects than the adult brain (Refs. 17-19). The combined effects of nicotine and menthol in the developing brain make youth who smoke menthol cigarettes particularly vulnerable to the effects of menthol on nicotine dependence.
Data from multiple studies across different populations and time periods demonstrate that menthol cigarettes contribute to greater nicotine dependence in youth and young adults1 than non-menthol cigarettes (Refs. 20-28). Menthol is a significant contributor to experimentation and progression to regular cigarette smoking among this population (Refs. 25, 29-31, 8). This is of particular concern since the vast majority of smoking initiation occurs during adolescence (Refs. 32, 8, 31, 33) and youth and young adults are more likely to try a menthol cigarette as their first cigarette than a non-menthol cigarette (Refs. 8, 31, and 33).
… Prohibiting menthol as a characterizing flavor in cigarettes would help to decrease the nicotine addiction resulting from menthol cigarette use, and thereby, decrease disease and death. (page 11-13)
FDA goes on to detail the underlying biological mechanisms for these effects:
2. Menthol Enhances Nicotine Addiction in the Brain
Menthol enhances the effects of nicotine in the brain by affecting mechanisms involved in nicotine addiction. Nicotine is the primary chemical in tobacco products that causes addiction through its psychoactive and reinforcing effects (Ref. 114). Nicotine addiction occurs as the result of repeated exposure to nicotine, which induces changes in the brain (Refs. 115, 9, and 116). Addiction to nicotine can lead to symptoms of nicotine dependence, which may include tolerance to the effects of nicotine, withdrawal symptoms upon cessation of use, and craving cigarettes (Refs. 9 and 1).
Upon inhaling smoke from a burning cigarette, nicotine is absorbed into the lungs and rapidly travels to the brain. Once in the brain, nicotine produces its initial effects by binding to nicotinic receptors, the primary targets for nicotine in the brain, and inducing release of the chemical dopamine (Refs. 115 and 9). Dopamine plays a major role in the pleasurable and reinforcing effects of smoking that promote continued use (Refs. 115 and 9). After repeated exposure to nicotine, nicotinic receptors become less responsive, prompting an increase in the number of brain nicotinic receptors; this process has been implicated in the development of nicotine addiction (Ref. 9).
A clinical study that analyzed brain images of adult non-smokers, menthol smokers, and non-menthol smokers found that menthol cigarette smokers have higher levels of brain nicotinic receptors than non-menthol smokers (Ref. 14). Studies in rodents have been used to provide insight into a mechanism for how menthol produces this effect in the brains of smokers. The nicotinic receptor composition, distribution, and function in the rodent brain is comparable to that of humans, and rodents can be trained to perform a variety of behavioral tasks (Refs. 117-119). Therefore, rodents serve as an appropriate model to examine the behavioral effects of nicotine and the effects of nicotine in the brain.
Studies demonstrate that menthol, like nicotine, binds to nicotinic receptors in the brain (Refs. 15 and 16), and menthol alone can increase the number of nicotinic receptors in the brain (Refs. 10 and 11). Consistent with clinical findings in menthol smokers (Ref. 14), animal studies also demonstrate that menthol in combination with nicotine increases the number of nicotinic receptors in the brain to a greater extent than nicotine alone (Refs. 10-12). This effect in the brain was accompanied by greater intensity of nicotine withdrawal signs in rodents treated with nicotine and menthol compared to those treated with nicotine alone (Ref. 10). Menthol also enhances nicotine’s effects on dopamine in the rodent brain. Animal studies demonstrate that nicotine-induced dopamine release is greater in the presence of menthol (Ref. 13). Additionally, menthol enhances nicotine-induced increases in dopamine cell activity to a greater extent than nicotine alone; these changes were associated with differences in behavioral responses to the rewarding effects of nicotine, where menthol-treated rodents exhibited greater reward for nicotine than those treated with nicotine alone (Ref. 12). These findings demonstrate that menthol’s effects on nicotine in the brain are associated with behaviors indicative of greater addiction to nicotine.
In combination with menthol’s flavor and sensory effects, menthol’s interaction with nicotine in the brain plays a role in making it easier to experiment, progress to regular smoking and dependence, and harder to quit smoking. (Page 35-37)
After going to the biology on the differences between adolescent and adult brains (page 36 to page 38) the FDA concludes, “Therefore, due to the combined effects of nicotine and menthol in the developing brain, youth who smoke menthol cigarettes are particularly vulnerable to the effects of menthol on progression to regular use and dependence.”
FDA goes on to explain how these biological effects result in “menthol facilitates experimentation and progression to regular cigarette use among youth and young adults,” (page 38) and “menthol contributes to nicotine dependence in young people” (page 39), and “menthol in cigarettes makes quitting smoking more difficult” (page 42).
After going through the epidemiological studies that show that menthol makes it harder to quit, FDA explains the underlying biology in the section on how “menthol’s interaction with nicotine in the brain makes it harder to quit smoking.” This section concludes:
Clinical and animal studies show that menthol enhances brain nicotinic receptor levels to a greater extent than nicotine alone (Refs. 14, 10, and 11). These changes occur in brain regions involved in the development of nicotine addiction (Refs. 10-12). Therefore, menthol’s ability to enhance the effects of nicotine in the brain contributes to why menthol smokers have greater difficulty quitting smoking compared to non-menthol smokers. (page 48)
These changes all depend on the presence of nicotine and menthol in the brain. How the nicotine and menthol were delivered doesn’t matter.
FDA recognizes that the tobacco industry understands these effects and uses that understanding to design its products:
Evidence from tobacco industry documents indicates that the industry has been adding menthol to cigarettes because of perceptions among new users that menthol cigarettes are less harsh and easier to smoke (Ref. 7). These documents indicate that menthol has traditionally been added to cigarettes as a design feature to attract new youth and young adult smokers (Refs. 7 and 6). For example, a 1987 document from one company states: “Menthol brands have been said to be good starter products because new smokers appear to know that menthol covers up some of the tobacco taste and they already know what menthol tastes like, vis-à-vis candy” (Ref. 112). Additionally, a 1978 document about a traditionally menthol-only cigarette brand states that the brand is “being purchased by Black people (all ages), young adults (usually college age), but the base of our business is the high school student” (Ref. 113). Menthol cigarettes continue to be used disproportionately by youth and new smokers (Ref. 44). (page 34-35)
Other industry research [11] shows that menthol is not only used in cigarettes as a flavor additive; tobacco companies know that menthol also has sensory effects and interacts with nicotine to produce tobacco products that are easier to smoke, thereby making it easier to expose smokers, especially those who are new and uninitiated, to the addictive power of nicotine.
Note that all of these effects are due to the chemical menthol rather than any combustion products of menthol or any aspect of the way a cigarette generates the nicotine/menthol aerosol that the user inhales.
There is no reason to expect that inhaled menthol delivered in an aerosol form where the aerosol is generated using a different technology (i.e., by heating up e-liquid or in a mini cigarette used in a heated tobacco product) would have any different biological or behavioral effects.
The bottom line
The evidence FDA musters in support of banning menthol in cigarettes applies equally to e-cigarettes and other inhaled tobacco products that FDA has authorized. FDA should immediately deny marketing orders for menthol e-cigarettes as well as drop the proposed exceptions in the menthol cigarette rule.
Unlike for cigarettes which requires a years-long rule making process, all FDA needs to do to stop menthol e-cigarettes is deny the PMTA applications.
[1] National Academies of Sciences, Engineering, and Medicine report, “Public Health Consequences of E-Cigarettes.” Washington, DC: The National Academies Press, 2018. Available at https://doi.org/10.17226/24952.
[2] Physicians for a a Smoke-free Canada. Science has marched on: it’s time to update the advice to Canadians. 2022. https://gem.godaddy.com/p/09d6831?pact=34037-166639209-13033929154-93fd8f1b153d5d6f1c70653de4f490c5472e45e6 . This blog post is an excellent summary of the evidence published after the NASEM report was prepared.
[3] Levy, D.T., R. Meza, Z. Yuan, et al. “Public Health Impact of a US Ban on Menthol in Cigarettes and Cigars: A Simulation Study.” Tobacco Control, 2021. Available at https://doi.org/10.1136/tobaccocontrol-2021-056604
[4] Wang, R.J., S. Bhadriraju, and S.A Glantz. “E-Cigarette Use and Adult Cigarette Smoking Cessation: A Meta-Analysis.” Research and Analysis, 111(2), 2021. Available at https://dx.doi.org/10.2105%2FAJPH.2020.305999
[5] Hedman L, Galanti MR, Ryk L, Gilljam H, Adermark L. Electronic cigarette use and smoking cessation in cohort studies and randomized trials: A systematic review and meta-analysis. Tob Prev Cessat. 2021 Oct 13;7:62. doi: 10.18332/tpc/142320. PMID: 34712864; PMCID: PMC8508281
[6] U.S. Department of Health and Human Services. Smoking Cessation. A Report of the Surgeon General. Atlanta, GA: U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health, 2020. https://www.cdc.gov/tobacco/data_statistics/sgr/2020-smoking-cessation/index.html
[7] Osibogun O, Bursac Z, Maziak W. Longitudinal transition outcomes among adult dual users of e-cigarettes and cigarettes with the intention to quit in the United States: PATH Study (2013-2018). Prev Med Rep. 2022 Feb 28;26:101750. doi: 10.1016/j.pmedr.2022.101750. PMID: 35256929; PMCID: PMC8897625
[8] Chen R, Pierce JP, Leas EC, Benmarhnia T, Strong DR, White MM, Stone M, Trinidad DR, McMenamin SB, Messer K. Effectiveness of e-cigarettes as aids for smoking cessation: evidence from the PATH Study cohort, 2017-2019. Tob Control. 2022 Feb 7:tobaccocontrol-2021-056901. doi: 10.1136/tobaccocontrol-2021-056901. Epub ahead of print. PMID: 35131948.
[9] Barufaldi LA, Guerra RL, de Albuquerque RCR, Nascimento A, Chança RD, de Souza MC, de Almeida LM. Risk of smoking relapse with the use of electronic cigarettes: A systematic review with meta-analysis of longitudinal studies. Tob Prev Cessat. 2021 Apr 27;29:29. doi: 10.18332/tpc/132964. PMID: 33928198; PMCID: PMC8078138
[10] Levy, D.T., C.J. Cadham, L.M. Sanchez-Romero, et al. “An Expert Elicitation on the Effects of a Ban on Menthol Cigarettes and Cigars in the United States.” Nicotine & Tobacco Research, 23(11): 1911-1920, 2021. Available at https://doi.org/10.1093/ntr/ntab121.
[11] Yerger VB. Menthol’s potential effects on nicotine dependence: a tobacco industry perspective. Tob Control. 2011 May;20 Suppl 2(Suppl_2):ii29-36. doi: 10.1136/tc.2010.041970. PMID: 21504929; PMCID: PMC3088468.

Stan, Has anyone done any studies on the effects of SECONDHAND vaping smoke? Thanks.
LikeLike
There are studies measuring levels of secondhand aerosol in the air and also showing that it is absorbed into bystanders’ bodies. I am not aware of any specific studies on health effects of secondhand aerosol exposure.
LikeLike