The 2009 Family Smoking Prevention and Tobacco Control Act gave the FDA jurisdiction to regulate tobacco products, defined as products “made or derived from tobacco.” This means that any product that includes nicotine derived from tobacco is subject to FDA jurisdiction. There has been discussion in academic circles about nicotine delivery products being made fromContinue reading “FDA should take Puff Bar at its word that it is not a tobacco product … and ban them as illegal drug delivery devices”
Author Archives: Stanton Glantz
Altria moving toward entering US marijuana market
Altria (owner of Philip Morris) has made two moves to get ready to enter the US marijuana market. It registered to lobby on marijuana commercialization in Virginia and has submitted two US patents for cannabis vaporization devices. According to the report in Cannabis Wire, “Altria supports the federal legalization of cannabis under an appropriate regulatoryContinue reading “Altria moving toward entering US marijuana market”
Smoke-free laws and cigarette taxes impact smoking behavior trajectories in youth and young adults
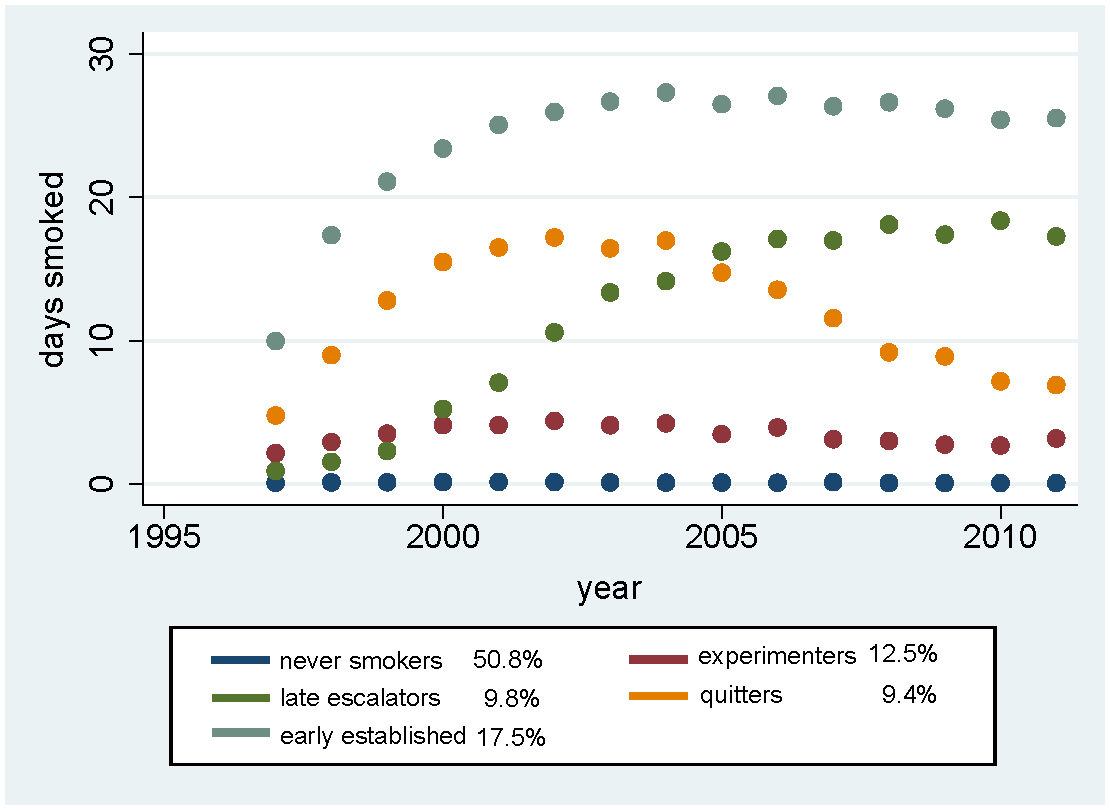
All smokers’ behavior is not the same. We and others has identified five trajectories of smoking behavior over time among youth and young adults based on demographic characteristics (graph above). Now Dorie Apollonio, Lauren Dutra and I have expanded this work to include the effects of changing tobacco control policies — smokefree laws and taxesContinue reading “Smoke-free laws and cigarette taxes impact smoking behavior trajectories in youth and young adults”
More opioid industry documents being made public
Just as litigation against the tobacco companies beginning with the Minnesota settlement has led to over 90 million pages of previously secret tobacco industry documents being made public, the recent settlement between state attorneys general and with McKinsey Company will make internal McKinsey documents available to the public. The tobacco industry documents have led toContinue reading “More opioid industry documents being made public”
Biden facing moment of truth on menthol
Under pressure from the tobacco companies Congress exempted menthol from the ban on “characterizing flavors” in cigarettes and other regulated tobacco products when it passed the 2009 Family Smoking Prevention and Tobacco Control Act that gave FDA authority to regulate tobacco products. During the negotiations a group of former Secretaries of Health and others criticizedContinue reading “Biden facing moment of truth on menthol”
You can watch the recorded UCSF Tobacco Center Billion Lives Symposium online
The program is here and the whole symposium can be watched here.
Pam Ling appointed new director of UCSF CTCRE
On January 28, 2020, UCSF School of Medicine Dean Talmadge King announced that Pam Ling will be the new director of the UCSF Center for Tobacco Control Research and Education. As the dean says, UCSF did a national search and Pam was selected. This is a great choice! From the Dean DearContinue reading “Pam Ling appointed new director of UCSF CTCRE”
Lessons for today from tobacco industry efforts to open the Thai market in the 1980s
Kade Patanavanich and I recently published “Successful countering of tobacco industry efforts to overturn Thailand’s ENDS ban” that documented recent efforts by Philip Morris and other pro-tobacco interests to open the Thai market to electronic nicotine delivery systems (ENDS), which under Thai law includes both liquid e-cigarettes and heated tobacco products like PM’s IQOS. NowContinue reading “Lessons for today from tobacco industry efforts to open the Thai market in the 1980s”
US Preventive Services Task Force finds evidence on e-cigs as a medicine for quitting “insufficient”
I have written several times about the differences between e-cigarettes as a consumer product and as a cessation medicine. pointing out that population observational studies are relevant for e-cigarettes as a consumer product and randomized clinical trials (RCTs) are relevant for e-cigarettes as a smoking cessation therapy. Now the US Preventive Services Task Force, aContinue reading “US Preventive Services Task Force finds evidence on e-cigs as a medicine for quitting “insufficient””
Three years later the NASEM report on e-cigs is out of date
Physicians for a Smokefree Canada just released this blog post which is an excellent summary of how the state of knowledge has advanced substantially since the National Academies of Science, Engineering and Medicine released its report on e-cigarettes in 2018. While the report was a good summary of the evidence when it was prepared, weContinue reading “Three years later the NASEM report on e-cigs is out of date”