Last week Tobacco Control published two peer reviewed scientific critiques of FDA’s authorization for RJ Reynolds to sell Vuse Solo e-cigarettes as well as a commentary on the two papers. Both papers recommend FDA exercise its authority to withdraw authorization to sell Vuse Solo and the commentary because FDA did not demonstrate that marketing VuseContinue reading “Three new papers detail FDA errors in authorizing Vuse Solo”
Category Archives: FDA
FDA pushes back date for finishing PMTAs (again)
The convenience store trade publication CSP reported that the FDA has pushed the anticipated date for finishing making decisions on premarket tobacco product applications (PMTAs) — mostly e-cigarettes — again, this time to the end of 2023. According to CSP, The Food and Drug Administration is going to need until the end of 2023 toContinue reading “FDA pushes back date for finishing PMTAs (again)”
UCSF Tobacco Center Billion Lives Symposium available for online viewing
As in past years, the symposium is archived and available to watch for free here. This is the program: Keynote Panel: 20 Years of the Industry Documents: Past, Present and Future: Presentations by Faculty and Postdoctoral Scholars: Closing Remarks, Chris Shaffer, MS, UCSF Assistant Vice Chancellor and University Librarian The last several years’ symposia areContinue reading “UCSF Tobacco Center Billion Lives Symposium available for online viewing”
RJR non-menthol Newport cigs are “substantially equivalent” to RJR Newport menthol
There are three ways that a tobacco company can get authorization to sell a new tobacco product in the US. The way that has attracted a lot of attention lately is through a premarket tobacco product application (PMTA); all the e-cigarettes have to go through this process. There are also two easier processes: submit aContinue reading “RJR non-menthol Newport cigs are “substantially equivalent” to RJR Newport menthol”
RJR tries to slither around CA ban on sale of flavored tobacco products with Newport, too
RJ Reynolds, BAT’s US subsidiary, has released its third effort to keep selling flavored cigarettes with its new Newport cigarettes that offer “bold lasting flavor” now that it can’t sell menthol cigarettes. The problem with what Reynolds is doing is that the law prohibits the sale of flavored tobacco products, not just menthol flavored tobaccoContinue reading “RJR tries to slither around CA ban on sale of flavored tobacco products with Newport, too”
A big step forward: FDA’s assessment of individual menthol e-cigs lines up with the scientific evidence
Two important memos dated October 25, 2022 detail internal FDA deliberations about how FDA considers specific menthol e-cigarettes in the context of the larger scientific literature on menthol. The two memos, one from the leadership of the Office of Science and another from Center for Tobacco Products Director Brian King and Deputy Director Michelle MitalContinue reading “A big step forward: FDA’s assessment of individual menthol e-cigs lines up with the scientific evidence”
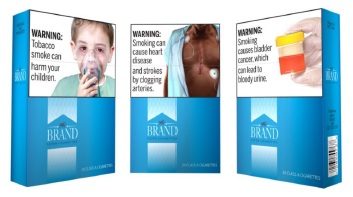
Texas Trump judge grasps at straws to reject graphic warning labels on cigarette packs; now the appeals can start
After sitting on the case for years, federal Judge J. Campbell Barker, an active member of the right-wing Federalist Society, finally issued his opinion vacating (rescinding) the FDA’s graphic warning labels on cigarette packs that Congress mandated in 2009 when it passed the Family Smoking Prevention and Tobacco Control Act. His decision rejecting the warningContinue reading “Texas Trump judge grasps at straws to reject graphic warning labels on cigarette packs; now the appeals can start”
Unredacted version of NJOY e-cig TPL shows what FDA is withholding from the public
When FDA authorized the sale of NJOY e-cigarettes in April 2022, it did not release the full Tobacco Product Lead (TPL) document that summarizes its scientific justification for doing so. In response to a Freedom of Information Act request for the full TPL, FDA inadvertently released an unredacted copy (details below). As of November 30,Continue reading “Unredacted version of NJOY e-cig TPL shows what FDA is withholding from the public”
After sitting on cigarette warning labels, judge announces he will rule on their legality (in a month)
Perhaps in response to the Department of Justice threatening to go over his head to the Count of Appeals, Texas Trump Judge J. Campbell Barker who has been sitting on the FDA’s Congressionally-mandated cigarette pack graphic warnings for years has announced the he expects to rule on the industry’s challenge to the warning labels byContinue reading “After sitting on cigarette warning labels, judge announces he will rule on their legality (in a month)”
RJR promotes “ice” cigarettes to get around Calif flavored tobacco sales ban
In addition to suing California on a last ditch attempt to block California’s ban on sale of flavored tobacco products that was overwhelmingly endorsed by the voters, RJ Reynolds (BAT’s US subsidiary) has sent mailers to smokers promoting new “non-menthol” cigarettes that taste like menthol (pictures above). Desmond Jenson from the Public Health Law CenterContinue reading “RJR promotes “ice” cigarettes to get around Calif flavored tobacco sales ban”