Last week Tobacco Control published two peer reviewed scientific critiques of FDA’s authorization for RJ Reynolds to sell Vuse Solo e-cigarettes as well as a commentary on the two papers. Both papers recommend FDA exercise its authority to withdraw authorization to sell Vuse Solo and the commentary because FDA did not demonstrate that marketing Vuse Solo would be “appropriate for the protection of the public health,” the legal standard that FDA is required to meet.
The key problems Lauren Lempert and I identified with FDA’s decision-making included:
(1) not adequately considering Vuse’s popularity with youth and evidence that e-cigarettes expanded the nicotine market and stimulate cigarette smoking;
(2) trading youth addiction for unproven adult benefit without quantifying these risks and benefits;
(3) not considering design factors that appeal to youth;
(4) not addressing evidence that e-cigarettes used as consumer products do not help smokers quit and promote relapse in former smokers;
(5) not discussing evidence that dual use is more dangerous than smoking;
(6) narrowly focusing on the fact that e-cigarettes deliver lower levels of some toxicants without addressing direct evidence on adverse health effects;
(7) downplaying significant evidence of other substantial harms;
(8) not acting on FDA’s own study showing no all-cause mortality benefit of reducing (but not stopping) cigarette use;
(9) improperly considering e-cigarettes’ high abuse liability and potential for high youth addiction and undermining tobacco cessation.
Andrew Meshnick, Lauren Faricy, and Boris Lushniak also made several of the same points, while going into more details about FDA’s failure to consider dual use, the most likely outcome of authorizing the sale of Vuse Solo, and additional issues FDA ignored regarding youth:
- FDA does not assess if the reduced toxin exposure to smokers who switch completely to Vuse outweighs the additional toxin exposures in the anticipated 95% of Vuse users who also continue cigarette smoking.
- The health effects studies FDA reviewed were small and contained largely healthy subjects, further obscuring the potential health impacts on those with chronic conditions like chronic obstructive pulmonary disease.
- There is no reason for FDA to assume that lack of evidence of risk is the same as evidence of no-risk. On the contrary, emerging evidence suggests increased risk of cardiovascular complications and stroke for dual users
- FDA limited its analysis to only current smokers and not the potential long-term poor health outcomes for youth who would otherwise not smoke cigarettes but who would use e-cigaretts
- FDA does not address pregnant users despite e-cigarettes being popular among young people who may become pregnant.
- The abuse liability analysis did not include youth, who are at an elevated risk of nicotine addiction due to their greater neuroplasticity and decreased impulse control compared with adults.
- FDA did not evaluate the risk of Vuse-induced adverse health effects in youth non-users.
- FDA did not address the evidence that e-cigarettes increase the total number of youth tobacco users beyond those otherwise likely to initiate combustible cigarettes alone.
(Boris Lushniak served as Acting Surgeon General among other important public health positions in government before moving to academia as dean of the University of Maryland School of Public Health.)
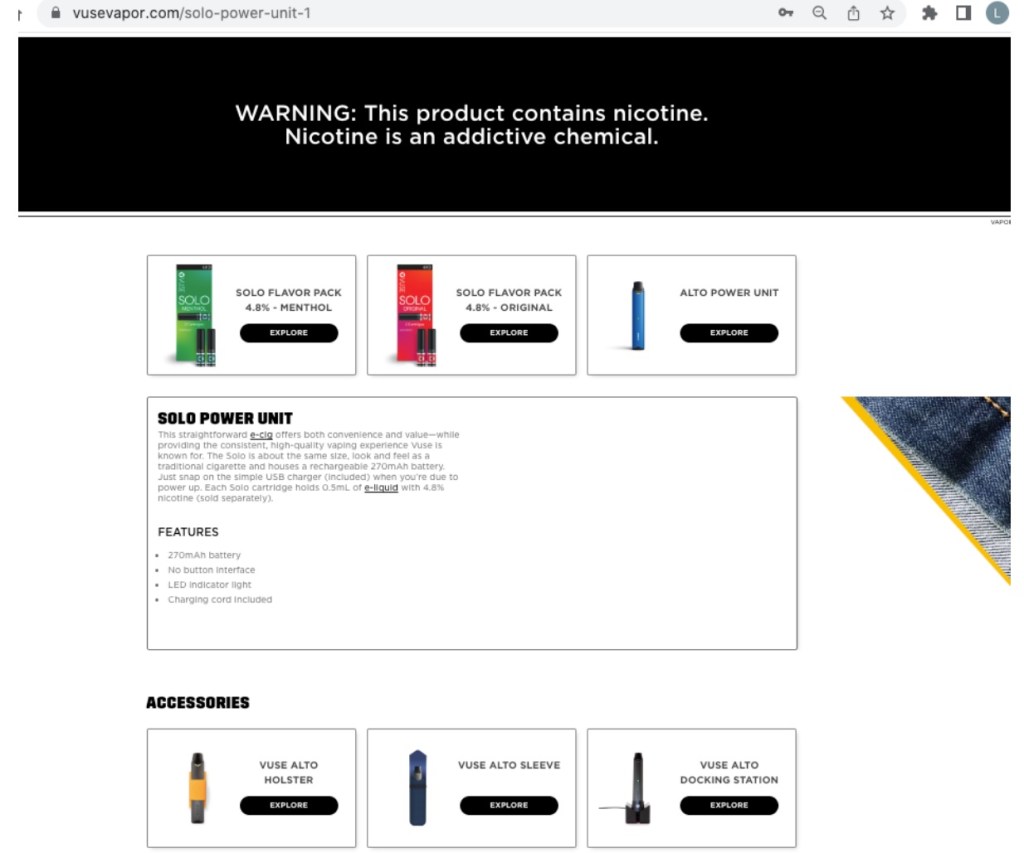
Both papers focused on health issues; neither commented in any detail on RJR’s marketing plans, another important aspect of authorizing the sale of new tobacco products. Meshnick and colleagues did note that FDA only suggested rather than requiring restrictions on RJR marketing practices. Although FDA’s decision relied in large part on its assessment that youth are not attracted to tobacco-flavored products, FDA failed to address the reality that Vuse Solo Menthol is marketed alongside Vuse Solo Original, and all Vuse Solo products are marketed together with Vuse Alto e-cigarettes, which come in a variety of flavors and are especially appealing to adolescents. Here is an example from RJR’s Vuse website (accessed 31 Feb 2023).
Eric Lindblom’s commentary on the two papers begins with noting that the problems with the FDA’s justification for authorizing the sale of Vuse Solo identified in the two papers “are too numerous to list here, but Glantz and Lempert and Meshnick et al reasonably conclude that FDA should withdraw its order allowing the marketing of the Vuse Solo e-cigarettes because the agency did not adequately evaluate the Vuse Solo application, did not establish that allowing its marketing was ‘appropriate for the protection of the public health’ and did not structure its final pre-market tobacco product application (PMTA) order to avoid increases in youth product use or unnecessary harms and risks.”
Lindblom moves beyond highlighting the problems with FDA’s decision-making for Vuse Solo and makes several suggestions about how FDA could use its existing ponderous processes combined with enforcement discretion to have positive health impacts beyond decisions about individual products:
FDA takes and enormous amount of time to review applications, make decisions and issue final PMTA orders, regularly missing statutory or court-ordered deadlines. That means many e-cigarettes have been left on the market, largely unregulated, despite clearly being inappropriate for the protection of the public health. Much of this problem comes from FDA largely failing to take action to remove entire categories of not-appropriate-for-the-protection-of-the-public-health e-cigarettes from the market but doing detailed, time-consuming case-by-case reviews of each pending application, instead. …
With its October 2021 order allowing the marketing of tobacco-flavoured Vuse Solo e-cigarettes, FDA also issued marketing denial orders for all Vuse Solo e-cigarettes with added flavours other than menthol. Accordingly, FDA could have immediately used its enforcement discretion to take off the market all other e-cigarettes still on the market with pending applications that also had added flavours other than tobacco or menthol—unless or until FDA made a decision on their pending application to allow their marketing. Instead, FDA left all those e-cigarettes on the market with those added flavours that FDA had formally found to strongly attract youth and increase youth use.
Similarly, FDA’s PMTA marketing order for the Vuse Solo tobacco-flavoured e-cigarettes included some marketing restrictions, such as requiring strict age and ID verification for any digital advertising or sales to reduce youth exposure and access and prohibiting ads on TV or radio programmes with 15% or more of their audiences being under the age of 21 years. Accordingly, FDA could have immediately announced that it would pull off the market any e-cigarettes on the market with pending applications for PMTA orders if they did not immediately begin complying with those same marketing restrictions.
As FDA had formally found those marketing restrictions necessary to make the marketing of tobacco-flavoured e-cigarettes appropriate for the protection of the public health, they were certainly necessary to regulate the marketing of other flavoured e-cigarettes as well. Following that same logic, FDA could have also taken action to issue a new final rule to apply those same restrictions to all other tobacco-nicotine products, or at least all those are or might be as or more harmful than e-cigarettes.
But FDA did not do either of these things.
Not only do these papers point to problems with the Vuse Solo decision (and subsequent authorizations of other e-cigarettes following the Vuse Solo model), but they provide a roadmap for FDA to modernize its approaches to more accurate, effective, and efficient scientific assessment of PMTAs. Let’s hope that Brian King, the first scientist to lead the Center for Tobacco Products, moves quickly to remedy these problems.
Here are the publication details:
Lindblom EN. How might FDA fix this e-cigarette PMTA mess? Commentary on Glantz and Lempert and Meshnick et al. Tob Control. 2023 Feb 9:tc-2022-057872. doi: 10.1136/tc-2022-057872. Epub ahead of print. PMID: 36764684. It is available for free here.
Glantz S, Lempert LK. Vuse Solo e-cigarettes do not provide net benefits to public health: a scientific analysis of FDA’s marketing authorisation. Tob Control. 2023 Feb 9:tobaccocontrol-2022-057296. doi: 10.1136/tc-2022-057296. Epub ahead of print. PMID: 36764683. It is available here.
Here is the abstract:
In October 2021, the US Food and Drug Administration (FDA) authorised marketing of RJ Reynolds Vapor Company’s (RJR) Vuse Solo e-cigarette through FDA’s Premarket Tobacco Product Application (PMTA) pathway. FDA concluded that RJR demonstrated Vuse products met the statutory standard of providing a net benefit to public health. A review of FDA’s scientific justification reveals deficiencies: (1) not adequately considering Vuse’s popularity with youth and evidence that e-cigarettes expanded the nicotine market and stimulate cigarette smoking; (2) trading youth addiction for unproven adult benefit without quantifying these risks and benefits; (3) not considering design factors that appeal to youth; (4) not addressing evidence that e-cigarettes used as consumer products do not help smokers quit and promote relapse in former smokers; (5) not discussing evidence that dual use is more dangerous than smoking; (6) narrowly focusing on the fact that e-cigarettes deliver lower levels of some toxicants without addressing direct evidence on adverse health effects; (7) downplaying significant evidence of other substantial harms; (8) not acting on FDA’s own study showing no all-cause mortality benefit of reducing (but not stopping) cigarette use; and (9) improperly considering e-cigarettes’ high abuse liability and potential for high youth addiction and undermining tobacco cessation. Because marketing these products is not appropriate for the protection of the public health, FDA should reconsider its Vuse marketing order as statutorily required and not use it as a template for other e-cigarette PMTAs. Policymakers outside the USA should anticipate that tobacco companies will use FDA’s decision to try to weaken tobacco control regulation of e-cigarettes and promote their products.
Meshnick AB, Faricy LE, Lushniak BD. Analysis of FDA’s Vuse market authorisation: limitations and opportunities. Tob Control. 2023 Feb 9:tobaccocontrol-2022-057540. doi: 10.1136/tc-2022-057540. Epub ahead of print. PMID: 36764685. It is available here.
Here is the abstract.
This special communication provides a physicians’ critique of the US Food and Drug Administration (FDA)’s decision to authorise the Vuse Solo (Vuse) Premarket Tobacco Application (PMTA). The PMTA authorisation represents the first time that FDA has authorised an Electronic Nicotine Delivery System (ENDS) for marketing in the USA. Using the FDA Decision Summary, the special communication identifies significant unanswered public health and scientific questions that prevent the authors from reaching FDA’s conclusion that Vuse meets the Appropriate for the Protection of the Public Health (APPH) standard. The authors recommend FDA suspend the market authorisation and use these questions to re-evaluate the PMTA, and to prospectively monitor whether Vuse meets APPH standard. The special communication advances the ENDS harm reduction conversation because it calls for national tobacco regulators to develop an epidemiological prediction of ENDS impact on the population and to expand the scope of their analysis to evaluate the impacts of ENDS on congenital birth defects, abuse liability and non-flavour drivers of youth usage. Through learning from the American experience regulating Vuse, national tobacco regulators around the globe will be better equipped to evaluate the impact of ENDS on the public health.
