Nicotine and Tobacco Research recently published four papers on menthol (menthol preference associated with greater nicotine dependence and greater urge reduction after smoking, biological effects of menthol on addiction. menthol use has not dropped among Black and Hispanic students, 85% of pregnant women smoke menthol and quit less) adding to the already-compelling case that menthol is a crucial additive that contributes to nicotine addiction and the health disparities that are increasingly evident in the tobacco epidemic.
These papers are accompanied by an excellent editorial, “Banning Menthol Cigarettes: A Social Justice Issue Long Overdue” by Christine Delveno, Ollie Ganz, and Renee Goodwin that details the politics of why key health advocated agreed to leave menthol out of the 2009 Family Smoking Prevention and Tobacco Control Act and efforts to keep it in. They highlight the consistent leadership the African American Tobacco Control Leadership Council has played on menthol from the beginning as well as the emergence of important new players exemplied by Parents Against Vaping e-Cigarettes (who have embraced the issue of flavors broadly, including products poor kids use like flavored little cigars).
They highlight the fact that, as the research base on the harms of menthol since 2009 has quintupled since 2009, the FDA has not addressed menthol (see figure from their paper).
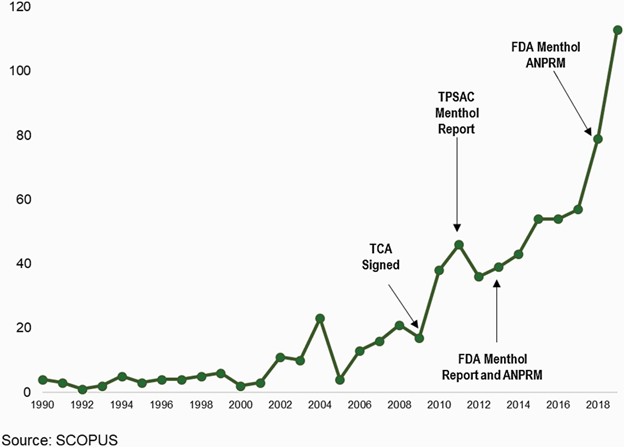
Abbreviations: ANPRM, Advance Notice of Proposed Rulemaking; TCA, Family Smoking Prevention and Tobacco Control Act; TPSAC, Tobacco Products Scientific Advisory Committee.
They also highlight that, while smoking non-menthol cigarettes has substantially dropped over time, menthol cigarettes have remained stable. And use has been concentrated not only among African Americans (85% of smokers smoke menthol), but also youth, LGBT, smokers with mental health problem and poor people.
The one point that Delnevo et al miss is that the FDA did, in fact, try to rein in menthol. The 2016 “deeming rule” that FDA submitted to the Obama White House had 17 pages on the adverse effects of flavors — including menthol — and prohibited their use in newly deemed tobacco products (including in e-cigarettes and little cigars, among other products) unless a tobacco company provided a specific justification that allowing use of a specific flavor was “appropriate for protection of public health,” the standard in the law.
Obama removed the 17 pages, leaving the door wide open to menthol and thousands of other flavors.
It is time for FDA to resuscitate those 17 pages and, after updating them to reflect that the case has more than doubled since 2016 (figure), and make another push with the White House.
On October 21, 2020, the California Tobacco Control Program launched www.WeAreNotProfit.org summing up the scientific and social justice case against menthol, including exposing how the tobacco companies have been fighting legislation to keep menthol in tobacco products.
Most important, cities and states should and will continue to pass comprehensive bans on sales of all flavored tobacco products, including menthol, to fill the void that the FDA continues to leave.

I agree a ban on menthol in smoked tobacco is long overdue and that politics have undoubtedly delayed action on this proposal. That said, the exclusive focus on characterizing flavors rather than more broadly on all flavor additives is something that public health groups in the US have overlooked. There are many additives in cigarettes and other smoked tobacco products that increase product appeal/attractiveness and inhalation of nicotine (thus, nicotine delivery and dependence).
Canada has a much more comprehensive approach to flavor/additive restrictions. The relevant prohibitions in sections 5.1 and 5.2 of Canada’s Tobacco Vaping Product Act (TVPA) can be found at https://laws.justice.gc.ca/eng/acts/T-11.5/page-2.html#h-449223
Information on the prohibited flavoring additives applicable to cigarettes in Schedule 1 of the TVPA can be found at: https://laws.justice.gc.ca/eng/acts/T-11.5/page-12.html#h-450049 ,specifically at items 1 and 1.2.
LikeLike