Yesterday (November 7, 2022), after Lauren Lempert finished working on our letter to the Regan-Udall Foundation raising issues about the FDA’s scientific standards for assessing premarket tobacco product applications, she browsed the Foundation page listing all the comments that had been submitted to the Expert Panel. The picture that emerges from looking at just aContinue reading “A troubling look inside FDA CTP staff highlighting interference with scientific assessment of PMTAs”
Category Archives: FDA
Recommendations to improve FDA premarket tobacco product application review to follow statutory mandates and improve scientific rigor
On October 21, 2022, Lauren Lempert presented at one of the Regan-Udall Foundation Expert Panels listening sessions on how to improve FDA operations of FDA’s Center for Tobacco Products operations. She and I followed up with a written comment letter detailing many of the problems with the tobacco product application processes, including both the premarketContinue reading “Recommendations to improve FDA premarket tobacco product application review to follow statutory mandates and improve scientific rigor”
Regan-Udall Foundation review of FDA tobacco regulation “listening sessions” dominated by pro-industry speakers
Regan-Udall Foundation review of FDA tobacco regulation “listening sessions” dominated by pro-industry speakers The FDA has asked the Regan-Udall Foundation for recommendations on how to improve tobacco product regulation. The Foundation’s Tobacco Independent Expert Panel has had three “listening sessions” that have been dominated by industry players and “harm reduction” advocates. I have previously expressedContinue reading “Regan-Udall Foundation review of FDA tobacco regulation “listening sessions” dominated by pro-industry speakers“
FDA pulls some menthol e-cigs off the market: What about the rest?
Last week FDA took the important step of denying permission for Logic Technology to market is menthol e-cigarettes. In its press release, FDA specifically recognized the attraction of menthol e-cigarettes to youth: For non-tobacco-flavored e-cigarettes, including menthol-flavored e-cigarettes, existing evidence demonstrates a known and substantial risk with regard to youth appeal, uptake and use. RecentContinue reading “FDA pulls some menthol e-cigs off the market: What about the rest?”
New report explains menthol marketing from the beginning and why we need to end it
Robert Jackler and his colleagues at the Stanford Research Into the Impact of Tobacco Advertising recently released “ADVERTISING CREATED & CONTINUES TO DRIVE THE MENTHOL TOBACCO MARKET: Methods Used by The Industry to Target Youth, Women, & Black Americans,” an encyclopedic history of menthol marketing in the United States. Jackler and his colleagues complement aContinue reading “New report explains menthol marketing from the beginning and why we need to end it”
Removing cannabis from Schedule I will make it possible to do long-needed research
On October 6, 2022, President Joe Biden announced pardons for people convicted to marijuana use and possession under federal law. He also announced that he was directing his administration to review marijuana’s listing as a Schedule I drug under the Controlled Substances Act, which is defined as one with a high potential for abuse, noContinue reading “Removing cannabis from Schedule I will make it possible to do long-needed research”
FDA and CDC release data highlighting FDA’s continuing failure to protect kids from ecigs
The FDA and CDC just published the first results from the 2022 National Youth Tobacco survey in the CDC’s MMWR. The 2022 survey shows that 1 in 4 youth use menthol e-cigs, further strengthening the case that FDA should end its de facto authorization of menthol e-cigs Specifically, 26.6% of kids say menthol is theirContinue reading “FDA and CDC release data highlighting FDA’s continuing failure to protect kids from ecigs”
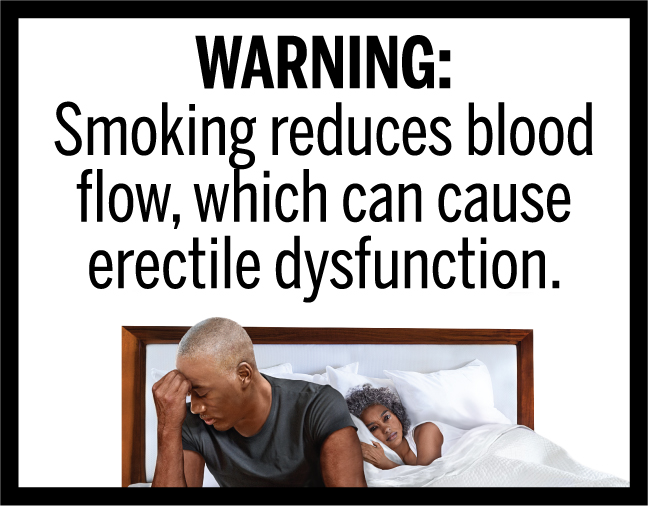
DOJ urges Trump judge who has been sitting on FDA cigarette pack warning labels to act
A Trump judge in Texas has been sitting on a case the cigarette companies filed against the FDA’s mandated health warning labels for two years. By not acting, he has effectively blocked the labels nationally but, by not ruling, there has been nothing for the Justice Department to appeal. The DOJ filing (below) is finallyContinue reading “DOJ urges Trump judge who has been sitting on FDA cigarette pack warning labels to act”
Trump judge continues to delay cigarette warning labels by doing nothing
Last May a federal judge in Texas appointed by President Trump again delayed ruling on the tobacco companies’ lawsuit against the Congressionally-mandated warnings on cigarette packs that the FDA finally issued in 2020. Now he has done it again, pushing the rule’s effective date for the warning labels by an additional 90 days, to OctoberContinue reading “Trump judge continues to delay cigarette warning labels by doing nothing”
Massachusetts’ comprehensive flavor ban cut menthol sales by 96% and overall tobacco sales by 25%
While the FDA continues to dither on getting rid of flavored tobacco products, most notably menthol e-cigarettes, states and localities continue to move forward. The tobacco companies continue to argue that such bans don’t work and that people will just switch to other products. Melody Kingsley, Hannah McGinnes, and their colleagues at the Massachusetts DepartmentContinue reading “Massachusetts’ comprehensive flavor ban cut menthol sales by 96% and overall tobacco sales by 25%”