My colleagues at UCSF and other universities and I have submitted this comment to the FDA in response to its proposal to prohibit the use of menthol in cigarettes. A PDF of the comment is here. The Regulations.gov tracking number is l6b-h3rv-v16n. FDA was correct in not quantifying consumer surplus in its analysis of theContinue reading “FDA was correct in not quantifying consumer surplus in its analysis of the proposed standard for menthol in cigarettes”
Author Archives: Stanton Glantz
FDA’s economic model underestimates the benefits of the proposed product standard prohibiting menthol in cigarettes, so the benefits would be even greater
My colleagues at UCSF and other universities and I have submitted this comment to the FDA in response to its proposal to prohibit the use of menthol in cigarettes. A PDF of the comment is here. The Regulations.gov tracking number is l6b-gxso-b6yq. FDA’s economic model underestimates the benefits of the proposed product standard prohibiting mentholContinue reading “FDA’s economic model underestimates the benefits of the proposed product standard prohibiting menthol in cigarettes, so the benefits would be even greater”
FDA should extend the scope of its proposed cigar flavor standard to prohibit flavors in all tobacco products, including e-cigarettes and smokeless tobacco, to make them less appealing to adolescents and young adults
My colleagues at the UCSF TCORS and I submitted this public comment to the FDA on its proposal to remove flavors from cigars. A PDF of the comment is here. The Regulations.gov tracking number is l6c-lwj1-vcu7. FDA should extend the scope of the proposed flavor standard to prohibit flavors in all tobacco products, including e-cigarettesContinue reading “FDA should extend the scope of its proposed cigar flavor standard to prohibit flavors in all tobacco products, including e-cigarettes and smokeless tobacco, to make them less appealing to adolescents and young adults”
FDA should not grant exemptions to its proposed standard banning menthol in cigarettes that would continue to allow menthol heated tobacco products or low nicotine cigarettes
FDA’s proposed standard banning menthol in cigarettes is very well-done except for its provision that would allow FDA to exempt heated tobacco products like Philip Morris’ IQOS, BAT’s glo or JTI’s Ploom — which are considered “cigarettes” under the law — or low nicotine cigarettes. (FDA has only authorized sale IQOS so far.) This isContinue reading “FDA should not grant exemptions to its proposed standard banning menthol in cigarettes that would continue to allow menthol heated tobacco products or low nicotine cigarettes”
FDA should prohibit all menthol flavor additives, compounds, constituents, and ingredients in cigarettes, and should not limit the proposed standard to prohibiting menthol as a “characterizing flavor”
My colleagues at UCSF and other universities and I have submitted this comment to the FDA in response to its proposal to prohibit the use of menthol in cigarettes. A PDF of the comment is here. The Regulations.gov tracking number is l6b-ew3y-c1ye. FDA should prohibit all menthol flavor additives, compounds, constituents, and ingredients in cigarettes,Continue reading “FDA should prohibit all menthol flavor additives, compounds, constituents, and ingredients in cigarettes, and should not limit the proposed standard to prohibiting menthol as a “characterizing flavor””
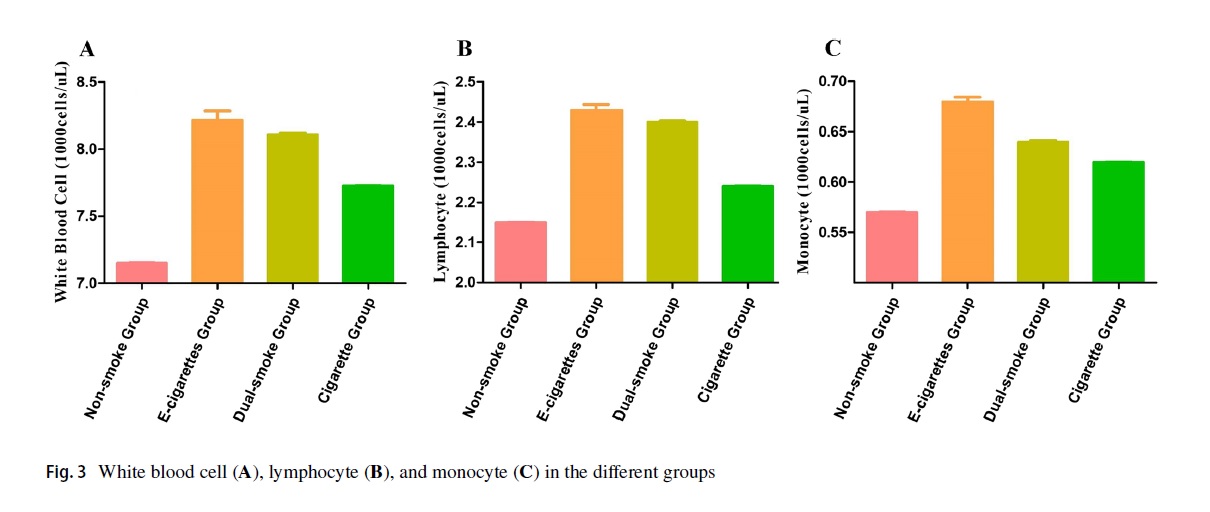
Another organ e-cigs adversely affect: blood
Yao Wang and colleagues compared the results of the Complete Blood Count (CBC), a routine blood tests used in clinical practice to diagnose a range of conditions, among participants in the US National Health and Nutrition Examination Survey (NHANES). In contrast to most national health survey, NHANES includes in person clinical tests on participants. TheirContinue reading “Another organ e-cigs adversely affect: blood”
Where are the Juul documents?
Perhaps the most important provision in the settlement between the State of North Carolina and Juul involved the release of previously secret internal Juul documents. These documents are particularly timely given the fact that Juul and the FDA are now fighting over FDA’s decision not to authorize the same of Juul e-cigarettes as well asContinue reading “Where are the Juul documents?”
FDA science leader Matt Holman leaves FDA for PMI
Matt Holman, who has served as head of the FDA Center for Tobacco Products since 2017, is leaving the FDA to take a job with Philip Morris International. According to a story in The Hill, a spokesperson for Philip Morris said Holman “is committed to helping existing adult smokers access scientifically substantiated smoke-free alternatives whileContinue reading “FDA science leader Matt Holman leaves FDA for PMI”
First epidemiological evidence linking e-cigs to cancer in people
E-cigarette advocates like to point out that because there is no combustion, e-cigarettes expose users to lower levels of many carcinogens (cancer-causing chemicals). Even so, there are still some carcinogens in e-cigarette aerosol exposure to e-cigarette aerosol damages DNA and reduces repair in animal and human cells and causes cancer in animals. Now there isContinue reading “First epidemiological evidence linking e-cigs to cancer in people”
Here’s an approachable introduction to the growing collection of industry insider documents on opioids, free to all
Caleb Alexander, together with colleagues at Johns Hopkins, UCSF and I just published “The Opioid Industry Documents Archive: A Living Digital Repository” in American Journal of Public Health. This paper describes the growing Opioid Industry Documents Archive that is part of the UCSF Industry Documents Library and outlines opportunities for research and public education withContinue reading “Here’s an approachable introduction to the growing collection of industry insider documents on opioids, free to all”